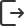STOCK CODE
600664
2025
Sustainability Report
HARBIN PHARMACEUTICAL GROUP CO. LTD.Contents
About Special Topic: Empowering with a Digital
About This Report ?? Message from the ?? Hapharm ?? 2025 Key Performance Sustainability
and Intelligent Engine Building a
Chairman in Sustainability ?? Management ??Group Foundation for High-Quality Develop-
??
ment Through Technological Innovation
Governance and Compliance to Product System for Value Creation Green Operations to Advance Employee Development to Build a
01 Solidify a Foundation for Stable 02 03 Low-Carbon Management 04 Safe and Shared FutureGrowth
Party Building Leadership and 23 Technological R&D and Innovation 39 Environmental Compliance 67 Employee Rights and Care 87
Responsibility Implementation Practices Management Talent Cultivation and Develop- 91
Construction of Corporate 26 Technology Ethics 43 Climate Change Tackling 68 ment
Governance System Industrial Cooperation and 44 Energy and Resource Manage- 72 Occupational Health and Safety 95
Internal Control and Compliance 31 Exchange ment Rural Revitalization Practices 101
Management Product Quality and Safety 45 Pollutant Emissions 77 Public Welfare and Social Action 103
Culture of Integrity and Honest 35 Management Waste Management 79
Operations Customer Rights and Interests 51 Ecosystem and Biodiversity 82
and Service Assurance Conservation
Supply Chain Management and 55
Security
Appendix
Information Security and Privacy 59
Key Performance Table 109 Protection
Index of Indicators 113 Medication Accessibility Enhance- 60
Reader Feedback Form 114 ment
Report Assurance Statement 116About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
About This Report Time Horizon
This Report covers the period from January 1 2025 to December 31 2025. To enhance the completeness and comparability of
the information some content may be appropriately traced back to previous years or extended beyond the reporting period
Report Explanation where necessary.
2025 Sustainability Report of Hapharm Group (hereinafter referred to as "this Report") is prepared and published by HARBIN Sources
PHARMACEUTICAL GROUP CO. LTD. (hereinafter referred to as "Hapharm Group" "the Company" or "we"). Building on the
Company's social responsibility disclosure efforts this is the first dedicated sustainability report that systematically integrates The data disclosed in this Report is primarily sourced from the Company's internal statistical data business management
and expands upon relevant information under the environmental social and governance (ESG) framework. It provides a focused systems and consolidated information from relevant functional departments. Some data is derived from audited financial
presentation of the Company's management philosophy key initiatives and progress in the field of sustainability for the year reports or third-party professional institutions. The Company adheres to the principles of authenticity accuracy and complete-
2025. This Report aims to facilitate more efficient comprehensive and objective communication and exchange with all stake- ness in data collection and consolidation to ensure the reliability of the information disclosed.
holders regarding the Company's sustainability work.How to Obtain This Report
Materiality This Report is published in electronic format and can be accessed and obtained from the website of the Shanghai Stock
Exchange (www.sse.com.cn) or the official website of Hapharm Group (www.hayao.com).This Report is available in both Chinese
The Company conducted a double materiality assessment on ESG-related topics for 2025 comprehensively identifying and and English versions. In case of any discrepancy between the two versions the Chinese version shall prevail.evaluating the impact of each topic on the Company's financial position stakeholders and the environment and society. For the
specific process and results of the double materiality assessment please refer to the "Double Materiality Analysis" chapter of Definitions
this Report.Quantitative Term Refers to Definition
For all quantitative indicators in this Report their measurement scope statistical methods relevant assumptions and the sourc- Hapharm Group the Company Refers to Harbin Pharmaceutical Group Co. Ltd.es of calculation tools or conversion factors used (if applicable) are explained accordingly to enhance the transparency and Harbin Pharmaceutical Group
the Group Refers to Harbin Pharmaceutical Group Holding Co. Ltd. (HPGC)comprehensibility of the information disclosed.Balance This Report Refers to 2025 Sustainability Report of Hapharm Group
Reporting period this year Refers to The period from January 1 2025 to December 31 2025
This Report strives to reflect the Company's management initiatives and phased achievements in the environmental social and
corporate governance fields in a true fair and comprehensive manner. It aims to avoid improperly influencing the judgment and Hayao General Factory Refers to Harbin Pharmaceutical Group General Factory
decisions of report users through selective disclosure information omission or improper presentation. Hayao No.6 Factory Refers to Harbin Pharmaceutical Group No.6 Pharmaceutical Factory
Consistency Sanchine Refers to Harbin Pharm Group Sanjing Pharmaceutical Co. Ltd.As this is the Company's first sustainability report prepared under the ESG framework the Company will unless otherwise speci- Sanjing Mingshui Refers to Harbin Pharmaceutical Group Sanjing Mingshui Pharmaceutical Co. Ltd.fied or in special circumstances continuously optimize and improve its statistical scope and disclosure methods for sustainabili- Shiyitang Chinese Medicine Refers to Harbin Shiyitang Chinese Medicine Co. Ltd.ty-related information disclosure based on existing disclosures. The Company will maintain relative consistency in disclosure
principles and methods to enhance the continuity and comparability of the information disclosed. Renmintongtai Refers to Harbin Pharmaceutical Group Renmintongtai Pharmaceutical Co. Ltd.Logistics Center Refers to Harbin Pharmaceutical Group Harbin Pharmaceutical Commercial Co. Ltd.Preparation Basis
Pharmaceutical Branch Refers to Pharmaceutical Branch of Hapharm Group Pharmaceutical Co. Ltd.This Report has been prepared in accordance with the Guidelines No. 14 of Shanghai Stock Exchange for Self-Regulation of Second Chinese Medicine Refers to Harbin Pharmaceutical Group Second Chinese Medicine Factory
Listed Companies – Sustainability Report (Trial) issued by the Shanghai Stock Exchange (SSE). It also references the Corporate Factory
Sustainability Disclosure Standards – Basic Standards (Trial) issued by nine departments including the Ministry of Finance of the Marketing Company Refers to Harbin Pharmaceutical Group Marketing Co. Ltd.People's Republic of China the Global Reporting Initiative (GRI) Standards issued by the Global Sustainability Standards Board
GNC China Refers to GNC China is jointly composed of GNC Hong Kong GNC Trading
(GSSB) IFRS S1 General Requirements for Disclosure of Sustainability-related Financial Information and IFRS S2 Climate-related Food Technology and Harbin Pharmaceutical Hong Kong II
Disclosures issued by the International Sustainability Standards Board (ISSB) the recommendations of the Task Force on GMP Refers to Good Manufacturing Practice
Climate-related Financial Disclosures (TCFD) and the United Nations Sustainable Development Goals (UN SDGs).GSP Refers to Good Supply Practice for Drugs
Reporting Scope GLP Refers to Good Laboratory Practice
RMB ten thousand yuan
hundred million yuan Refers to RMB yuan ten thousand yuan hundred million yuan
In principle this Report covers HARBIN PHARMACEUTICAL GROUP CO. LTD. and its subsidiaries included in the consolidated
financial statements. Where individual indicators do not cover all entities due to statistical scope business nature or data
availability corresponding explanations have been provided in the report.
03 04About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability
Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability ReportCreation
Chairman Group Sustainability Stable Growth Management Shared Future
Message from the Chairman
Focusing on present development to solidify foundations for long-term growth; looking ahead to drive
transformation and foster momentum through innovation.Over the past year the Company has continued to refine its modern corporate governance system treating compli-
ant operations and risk prevention and control as essential pillars of stable operations. Through a series of institu-
tional arrangements it has continuously enhanced decision-making transparency and management standardiza-
tion. At the same time the Company has steadfastly advanced technological innovation and digital
transformation—from optimizing its R&D system to implementing smart manufacturing practices and from quality
data governance to reengineering management processes—creating synergy between technology and manage-
ment to embed reform outcomes as organizational capabilities and drivers of development. The Company firmly
believes that only by building solid foundations and pursuing deep innovation can an enterprise maintain resilience
amid complex and changing environments and steadily advance toward longer-term development.Upholding quality as the cornerstone to honor its original mission and prioritizing responsibility to
protect health; building trust through dedicated service and demonstrating value through responsible
actions. The pharmaceutical industry carries the public's high expectations for safety and efficacy.The Company continues to strengthen its full-lifecycle quality management and pharmacovigilance systems
shifting risk identification and process controls upstream to the R&D and production stages. Through rigorous
quality process management it ensures that every medicine meets the test of standards and time. Concurrently
the Company continuously enhances customer service capabilities and medication accessibility by providing
professional pharmacy support and developing frontline services to better align health services with societal
needs. For Hapharm quality is not merely a baseline for survival but a solemn commitment to the public and the
fundamental basis for sustained and stable development.HARBIN PHARMACEUTICAL Embracing green trends to promote sustainable development and expanding horizons through syner-
GROUP CO. LTD. gistic collaboration; caring for employees to build collective strength and co-creating an ecosystem
for a shared future.Party Secretary and Chairman
In response to resource and environmental constraints the Company integrates green principles throughout its
production and operations continuously advancing energy conservation consumption reduction and environ-
mental protection to ensure more resilient and responsible development. Meanwhile the Company places great
emphasis on employee development and industrial collaboration fostering talent promoting industry-aca-
demia-research partnerships and engaging in social practices to drive mutual progress among the enterprise its
partners communities and the broader industry. The Company deeply understands that genuine sustainability is
a process in which enterprises and society mutually empower and share outcomes.As times evolve so do health needs; as the industry progresses responsibilities deepen. Looking ahead with steadfast resolve to create long-term value; walking hand-in-hand toward a shared
vision and co-writing a new chapter in health.Standing at a new stage of profound transformation in the pharmaceutical and healthcare sector the Company recognizes with Going forward sustainability will remain a critical benchmark for measuring corporate resilience and governance
increasing clarity that the value of a pharmaceutical enterprise lies not only in its scale and performance but also in its reverence maturity. Hapharm Group will leverage a stronger governance foundation a more determined pace of innovation
for life and health its commitment to social responsibility and its continuous investment in long-term sustainability. Hapharm and a more open approach to responsibility to translate sustainability principles into enduring value creation. The
Group has always upheld safeguarding public health as its founding mission closely integrating high-quality development with Company is committed to advancing alongside all stakeholders aligned in the mission of safeguarding public
social value creation. This year the Company is releasing its sustainability report for the first time under a systematic ESG frame- health ensuring that corporate growth and social progress reinforce each other and jointly crafting a new chapter
work. This represents both a comprehensive review of years of governance practices and developmental achievements and marks of high-quality development for Hapharm's future through accumulated trust and shared value over time.a significant upgrade in Hapharm's development philosophy management systems and expression of responsibility. It reflects the
Company's firm commitment to advancing high-quality development with greater transparency standardization and a long-term
orientation.
05 06About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
About Hapharm Group
Company Profile
The Company focuses on the pharmaceutical and healthcare industry primarily engaging in pharmaceutical research and develop- The Company’s pharmaceutical R&D and manufacturing business covers chemical drugs traditional Chinese medicine biologics
ment manufacturing wholesale and retail businesses. It is a large domestic high-tech pharmaceutical enterprise integrating R&D healthcare products medical devices and aesthetic medicine products. Its product portfolio focuses on five major therapeutic
manufacturing and sales. According to the industry classification standards issued by the China Association for Public Companies areas including cardiovascular and cerebrovascular diseases digestive and metabolic systems respiratory systems anti-infec-
the Company belongs to the pharmaceutical manufacturing industry (C27). The Company was listed on the Shanghai Stock tives and anti-tumor treatments with more than 500 products currently in production and on sale. Major products include Zinc
Exchange in June 1993 (Hapharm Group 600664.SH) becoming the first listed company in China’s pharmaceutical industry and Gluconate Oral Solution HAYAO Calcium-Iron-Zinc Oral Liquid Compound Calcium Gluconate Oral Solution Erythropoietin
the first listed company in Heilongjiang Province. Injection Amoxicillin Capsules Shuanghuanglian Oral Liquid Penicillin Sodium for Injection Qufeng Gutong Gel Patch Lacidipine
Tablets and Pediatric Paracetamol Artificial Cow-Bezoar and Chlorphenamine Maleate Granules.During its development the Company has continuously optimized its business structure and integrated resources. In 2015 the
Company reorganized its assets and business segments through asset restructuring establishing a pharmaceutical commercial The Company’s pharmaceutical wholesale and retail business is primarily conducted through its listed subsidiary Renmin Tongtai
listing platform (Renmin Tongtai 600829.SH) and forming a business layout in which pharmaceutical manufacturing and pharma- a well-known regional pharmaceutical distribution enterprise and the largest pharmaceutical commercial company in Heilongjiang
ceutical distribution operate relatively independently while developing in coordination. Province. Renmin Tongtai’s main businesses include pharmaceutical wholesale pharmaceutical retail pharmaceutical logistics
services and medical services. Its operations cover chemical drugs proprietary Chinese medicines and biologics as well as the
sale of medical devices Chinese herbal decoction pieces health foods daily necessities glassware and chemical reagents.
600664.SH
The Company was listed on the first listed company
Shanghai Stock Exchange in June becoming the first listed company in 600829.SH
1993 (stock code: 600664.SH) China's pharmaceutical industry and created Renmintongtai
the first listed company in Heilongji- (stock code: 600829.SH) as
ang Province. its listed platform for
pharmaceutical commercial
Over 500 activities.in-production and
Human Erythropoietin
Injection marketed
specifications Shuanghuanglian Oral Liquid
Compound Calcium Gluconate
Zinc Gluconate Oral Solution Oral Solution
GNC Vitapak Daily Nutrition Pack
Hayao Brand Calcium-Iron-Zinc
Oral Liquid
Lacidipine Tablets
Amoxicillin Capsules
07 08About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Company Honors 2025 Milestones
Brand and product market recognition
Sanjing Zinc Gluconate Oral Solution (Strawberry Flavor) obtained its
The Oral Liquid No. 1 Workshop of Sanchine and the filing certificate and completed the license expansion. This project
Solid Dosage Form No. 1 Workshop of Hayao No.6 received approval for a health food filing certificate marking the first-ever
Factory were recognized as "Heilongjiang Province approved zinc supplement in sachet format signifying that the Company's
CPHCF Award for Best-Selling West Lake Award for Most Advanced-Level Digital Workshops." R&D capabilities and product quality in the health industry have been
Products in China's Retail Popular Star Single Product 2025 Healthy China highly recognized by authoritative institutions.Sanjing Calcium Gluconate
Pharmaceutical Market in Pharmacies Brand List Calcium Lactate Oral Liquid (Peach Flavor) obtained its filing certificate
Harbin Pharm Group Sanjing Harbin Pharmaceutical Group Harbin Pharm Group Sanjing and completed the license expansion. As the Company's first calcium
Pharmaceutical Co. Ltd. No.6 Pharmaceutical Factory Pharmaceutical Co. Ltd. supplement in sachet format it represents a "zero-to-one" breakthrough in
The revitalization and industrial this field.upgrading project for the Xuefu Road
plant area of Hayao General Factory was
launched to transform it into an
"Industry+N" integrated industrial park.Acanthopanax Senticosus Base of
Innovation and market practice achievements Harbin Pharmaceutical Group Shiyitang
Co. Ltd. and Second Chinese Medicine
Factory under Harbin Shiyitang Chinese
Medicine Co. Ltd. successfully passed
the provincial good agricultural practice
First Batch of Famous Consumer (GAP) compliance inspection.Renmintongtai ranked 33rd on the "2024
Product Enterprise Brands in
Heilongjiang Province Top 100 Pharmaceutical Retail Enterprises
HARBIN PHARMACEUTICAL by Total Sales" list.GROUP CO. LTD.West Lake Award for West Lake Award for Three new products namely
Innovative Marketing Case Innovative Marketing Case Lactulose Oral Solution Oseltamivir
Harbin Pharmaceutical Group HARBIN PHARMACEUTICAL Phosphate Dry Suspension and Iron
Marketing Co. Ltd. GROUP CO. LTD. Polymaltose Complex Oral Solution
Industry Recognition and Collaborative Impact have obtained drug registration certificates. In addition two active
pharmaceutical ingredients
including Calcium Lactate API and
Iron Polymaltose Complex API have
been approved for marketing
application. This further enriches
the Company’s product portfolio
and continuously promotes the
construction of its product pipeline.CPHCF Award for Outstanding
Fulfillment Chain Enterprise in Partner in the Pharmaceutical Silver Award in the Medical and
China's Pharmaceutical Retail Manufacturing Sector Health Communication Category
Renmintongtai Pharmaceutical HARBIN PHARMACEUTICAL HARBIN PHARMACEUTICAL
Chain Co. Ltd. GROUP CO. LTD. GROUP CO. LTD.
1Among them Iron Polymaltose Complex Oral Solution obtained the drug registration certificate in January 2026 and Iron Polymaltose Complex API was
approved for marketing application as a chemical active pharmaceutical ingredient in January 2026.
09 10About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
2025 Key Performance in Sustainability
Economic performance Governance performance Social performance
Operating revenue of RMB Independent directors accounted Total R&D investment: Total number of valid patents held: 14 responsible marketing
15.964 billion for 33.3% 203.5182 million RMB 220 audits
Performed 761 supplier DTP pharmacies served a cumulative Total number of employees:
Total assets: RMB Percentage of female directors: training sessions total of 250000person-times 10039 persons
14.288 billion 44.4%
Number of people assisted
Total employee training duration: Employee trade union coverage
Net profit attributable to sharehold- 248712.2 hours rate: 100 through the Company's public %
welfare initiatives: 5682
ers of the listed company: RMB 9 training sessions for directors
0.362billion
Basic earnings per share: RMB
0.14 per share 14 investor relations activities
Environmental performance
Net assets attributable to share-
100% integrity risk assessment coverage
holders of the listed company: RMB
Total environmental management Total environmental protection investment:
5.688 for the Company and its subsidiariesbillion
training duration: 3569 hours RMB 15.8187 million
Total tax payment: RMB 100% employee signing rate for compli-
Photovoltaic power generation: Percentage of waste reclaimed and reused:
0.733billion ance commitment letters 4784.65 MWh 91.42%
11 12About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon Creation to Build a Safe and
Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Management Shared Future
Stakeholders Topics of concern Communication channels
Sustainability Management
Innovation-driven development
Information disclosure
Compliant operation
Investor hotline
Internal control and risk management
Sustainability Management System Investor communication platformCorporate governance
Shareholders and On-site/telephone research
investors Shareholder rights protection Social media
Climate change tackling
Aligning with its governance structure and operational realities the Company has established a sustainability management frame-
work led by the Board of Directors with participation from management and coordinated implementation by relevant departments
to research advance and supervise sustainability-related matters.Information disclosure
Investor hotline
Compliant operation
Investor communication platform
As the coordinating management body for the Company's sustainabili- Creditors Internal control and risk management Offline meetings
Strategy and Sustainability ty work it is responsible for overall supervision and guidance of major Social media
Committee sustainability-related matters deliberating key topics and submitting
decision-making recommendations to the Board of Directors.Employees Employee satisfaction surveys
Undertake support and coordination responsibilities for the
Office of the Board of Corporate governance Internal communication platform
Directors Committee's daily operations and ensure the orderly advancement Company employees Energy usage Employee training
of sustainability-related topics.Implement sustainability requirements in their respective domains Information disclosure
Relevant according to assigned responsibilities advance measure implementa- Industry cooperation and exchange Event participation
business and
functional tion and provide relevant information data and execution feedback to Partners and industry
Anti-unfair competition Offline meetings
departments establish a collaborative advancement mechanism. associations Social media
Hapharm Group's sustain-
ability governance structure
Information disclosure
Safety and quality of products and services Customer service hotline
Customer rights and interests protection Customer satisfaction surveys
In alignment with sustainability-related management requirements and ESG development needs the Company organizes special- Responsible marketing Marketing activities
ized ESG training to continuously enhance the overall awareness and understanding of sustainability topics among relevant Customers Data security and customer privacy protection After-sales service
management personnel and business units. The training further strengthens compliance and responsibility awareness regarding Social media
environmental social and governance matters laying a foundation for subsequent sustainability work and information coordina-
tion.Circular economy
Supply chain security Supplier communication and exchange
Equal treatment to small and medium-sized Supplier training and seminars
During the reporting period the Company Suppliers and contractors enterprises
Medical accessibility and inclusiveness
Organized ESG training for ?? participants Ecosystem and biodiversity conservation Information disclosure
Pollutant emissions
Social media
Contributions to the society
Media and the public Event participationWaste disposal
Usage of water resources
Communications with Stakeholders
Daily communication
The Company values communication with all stakeholder groups. Focusing on business operations and sustainability-related Due diligence Offline meetings
topics it continuously monitors reasonable expectations across stakeholder groups and engages through multiple channels. Environmental compliance managementGovernment and Training participation
Feedback received serves as a key input for improving management and optimizing decision-making supporting the Company's regulatory agencies Rural revitalization Information disclosure
stable operations and long-term development.
13 14About This Message About 2025 Key Hapharm Performance in Sustainability
Governance and Compliance Green Operations to Employee Development
Report from the Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Material Topic Matrix
High
In advancing its sustainability management the Company systematically identifies sustainability-related challenges and opportu-
nities in its operations by considering key capital market concerns national policy directions industry trends and its own strate-
gic priorities. On this basis it has established and refined a materiality assessment mechanism for sustainability topics evaluating
them along two dimensions—financial materiality and impact materiality—to determine priority levels. The assessment outcomes Access to Medicines and
serve as a critical basis for formulating management measures and optimizing decisions. Pharmaceutical InclusivenessCustomer rights and
interests protection
Safety and
Responsible quality of
marketing products and
?? Establishment of a
services
?? Stakeholder survey Supply chain securitysustainability topic library
Pollutant
emissions
Corporate governance
Requirements from regulators Distributing stakeholder survey Internal control and
Contributions risk management
capital markets and rating questionnaires; Data security and customer to the society Industry cooperation Employees
agencies; privacy protection and exchange
Gathering statistics on issues of Anti-unfair competitionEqual treatment to small and
Macro-policy and industry standard concern to stakeholders; medium-sized enterprises Innovation-driven Environmental compliance development
requirements; management
Organizing focus group interviews. Communications with Waste disposal stakeholders
Peer practices company business
?? Company management Rural revitalization Anti-commercial bribery and practices and characteristics. survey anti-corruption
Usage of water resources Energy usage
Shareholder rights protection
Distributing company management
survey questionnaires; Ecosystem and
biodiversity conservation Due diligence Climate change tackling
Analyzing the financial impact of Ethics of science and technology Compliant operation
sustainability topics; Circular economy
Organizing focus group interviews.?? Materiality assessment ?? Formation of a double Highmateriality matrix
Financial materiality
Assessing financial materiality; Forming a double materiality matrix Governance issues Environmental issues Social issues
to present the results of topic
Assessing impact materiality; analysis and assessment;
Determining the materiality The Board of Directors’ review of
threshold. the double materiality matrix.
1516
Impact materialityAbout This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Special Empowering with a Digital and Intelligent Engine Building a Foundation
Topic for High-Quality Development Through Technological Innovation
Focused on enhancing production governance capabilities and quality control the Company systematically advances digital
workshop construction embedding digital technologies throughout production operations and quality management. It continu- Controlled Production and Quality Throughout the
ously strengthens a production management foundation characterized by controllable quality visible processes and traceable Entire Process
data establishing digital manufacturing practices that meet regulatory requirements and offer replicable value.In production process management electronic batch records (EBR) serve as the core enabling systematic recording and audit
trail management of weighing material charging process parameters and critical operational nodes. It mitigates risks arising from
manual data entry and information dispersion greatly improves data integrity and traceability and effectively ensures stable and
Unified Architecture and System Integration controlled production processes. Product quality data is synchronized with production data allowing centralized analysis and trend identification through the quality management and analytics system. This supports deviation management non-conforming
product control and continuous improvement shifting quality management from post-event correction toward real-time process
monitoring and proactive early warning thereby continuously strengthening closed-loop quality issue management capabilities.The oral liquid formulation workshop of Sanchine and the oral solid dosage form workshop of Hayao No.6 Factory are advancing
digital upgrades under a unified technical architecture and management standards. Through coordinated system integration and
on-site retrofitting they are progressively building a digital operations system covering production execution quality control
equipment operation and energy management. At the system level both workshops have uniformly deployed supervisory control
and data acquisition (SCADA) systems with the manufacturing execution system (MES) as the core interconnected with the
equipment asset management (EAM) system warehouse management system (WMS) energy management system (EMS) and
quality retrospection and analysis system (QRS). This enables centralized collection and collaborative management of production
quality equipment and energy consumption data laying the groundwork for replication and scaled deployment across other
workshops.Digital production site at Sanchine Digital prod哈uc药tio六n 厂site车 a间t H数ay字ao 化No生.6产 Fa现cto场ry workshop
Digital Management System Platform Digital Exhibition & Display Platform
17 18About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability
Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Group Sustainability Stable Growth CreationChairman Management Shared Future
Special Empowering with a Digital and Intelligent Engine Building a Foundation
Topic for High-Quality Development Through Technological Innovation
Equipment Intelligence and Operational Efficiency
Improvement Capability Building and Demonstration Effectiveness
Hayao No.6 Factory continues to advance digital workshop construction through equipment configuration and automation While ensuring stable system operations the Company simultaneously advances digital capability building. Through internal
upgrades introducing AGVs collaborative robots and automated production lines and deploying intelligent equipment such as training and hands-on practice it cultivates versatile talent who understand both production processes and digital applications
multi-functional fluidized beds and high-speed tablet presses to drive the automation and standardization of key processes. A total enhancing the system's self-operation and continuous optimization capabilities to ensure long-term stable realization of digital
of 183 production units are under centralized monitoring with a 90% connectivity rate for critical equipment. Operational data outcomes. The relevant projects have received provincial-level digital workshop certification and national Level 3 Intelligent Manu-
analysis has reduced unplanned downtime by approximately 30% and increased overall equipment effectiveness (OEE) by an facturing Capability Maturity certification. While strengthening compliance management these initiatives ensure process stability
average of 10.5% significantly improving equipment stability and resource utilization efficiency. Based on visualization tools such and consistency through precise control and end-to-end traceability fundamentally enhancing the quality and safety of finished
as the production cockpit the real-time production status quality metrics and equipment performance are centrally displayed. pharmaceutical products—a contribution of significant importance to public medication safety and societal health.Operational efficiency in key processes has improved by 10%–22% material traceability time has been shortened by 40%–70%
order changeover efficiency has increased by approximately 80% inventory accuracy has reached 99.5% and inventory backlog
risk has been reduced by about 25%.Sanchine has introduced leading domestic oral dosage production equipment and through efficient integration achieved full-pro-
cess automation across washing drying filling sterilization visual inspection and packaging. By implementing SCADA and MES
systems it has enabled seamless data interconnectivity between equipment and systems achieving a 100% connectivity rate for
critical equipment and establishing a robust foundation for production monitoring. Replacing manual material handling with an
integrated system of automated conveyance tracks AGVs and mobile terminals has increased on-time material delivery in
workshops to 98% and delivery accuracy to over 99.5%. Through a dynamic process optimization model equipment OEE has
reached 80% and on-time order delivery has improved by 13%. Additionally energy collection and management systems have
enabled significant energy savings in production reducing energy consumption per RMB 10000 of output value by 15.9%.Certificate of Heilongjiang Province Advanced-Lev- Intelligent Manufacturing Capability
el Digital Workshop Maturity Assessment Certificate
Digital workshop management platform AGV logistics turnover at Sanchine
192001
Governance and
Compliance to Solidify
a Foundation for
Stable Growth
The content of this chapter
Party Building Leadership and Responsibility Implementation ??
Construction of Corporate Governance System ??
Internal Control and Compliance Management ??
Culture of Integrity and Honest Operations ??About This Message About 2025 Key Hapharm Performance in Sustainability
Governance and Compliance Green Operations to Employee Development
Report from the Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Party Building Leadership and Responsibility
Implementation Party Building-Led Key Project Initiatives
The Company insists on leveraging Party building to guide synergistic industry-academia-research collaboration and talent
system development treating joint Party building as a vital linkage for advancing scientific innovation technology transfer and
The Company adheres to the leadership of the Party in guiding its reform development and governance practices. Party building talent cultivation. Centered on actual enterprise needs and industrial demands it fosters co-construction partnerships with
work is conducted in strict accordance with the Constitution of the Communist Party of China Regulations on the Work of Primary primary-level Party organizations in universities and research institutes promoting organizational co-building resource sharing
Organizations of State-owned Enterprises of the Communist Party of China Regulations on the Election of Primary Organizations and mechanism alignment to seamlessly connect research capabilities industrial platforms and talent pipelines. Within the
of the Communist Party of China and other relevant rules. It continuously improves the operational mechanism of Party building framework of deep integration between Party work and core operations the Company explores practical pathways where joint
within a standardized and institutionalized framework and regards high-quality Party building as an important support for enhanc- Party building facilitates technological breakthroughs and collaborative mechanisms support talent development gradually
ing corporate governance capacity and operational efficiency. forming a working model of Party-led industry-academia-research-linked and talent-synergized advancement—providing
Focusing on the integrated development and mutual promotion of sustained support for high-quality development.Party building and business operations the Company integrates Party
building into the entire process of corporate governance production
operation reform and development. It keeps transforming the political
and organizational advantages of the Party into governance efficiency Case Party building-led practices in production efficiency improvement and lean management
and development momentum. In practice relying on the Four Promo-
tions mechanism of Party building promoting R&D production Hayao No.6 Factory targeting improved production efficiency and cost optimization uses the "Party Branch
marketing and services the Company embeds the functions of prima- Secretary Leads Projects" initiative as a key lever to deeply integrate Party building into production organization
ry Party organizations into core business links systematically. This and on-site management. Branch secretaries focus on critical areas—including solid dosage production process
achieves full-chain coordination between Party building and business optimization quality testing warehouse management and digital transformation—leading the establishment of
operations improves the pertinence and effectiveness of Party breakthrough projects and organizing core Party members into dedicated teams. Through lean production
leadership and provides solid support for the steady operation and management process streamlining and technological upgrades they continuously address operational bottle-
sustainable development of the enterprise. Party Member Training necks. These projects have achieved phased progress in shortening production cycles optimizing resource
allocation enhancing equipment utilization and advancing digital management demonstrating the positive role
of Party leadership in driving production efficiency and management upgrades.Party Building-Led Synergistic Development of Industry Academia
Research and Talent
The Company insists on leveraging Party building to guide synergistic industry-academia-research collaboration and talent
system development treating joint Party building as a vital linkage for advancing scientific innovation technology transfer and
talent cultivation. Centered on actual enterprise needs and industrial demands it fosters co-construction partnerships with
primary-level Party organizations in universities and research institutes promoting organizational co-building resource sharing
and mechanism alignment to seamlessly connect research capabilities industrial platforms and talent pipelines. Within the
framework of deep integration between Party work and core operations the Company explores practical pathways where joint
Party building facilitates technological breakthroughs and collaborative mechanisms support talent development gradually
forming a working model of Party-led industry-academia-research-linked and talent-synergized advancement—providing
sustained support for high-quality development.Hayao No.6 Factory's "Party Branch Secretary Leads Projects" Initiative
Award Ceremony for Inheriting the Scientists' Spirit and Advancing Innovation
23 24About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability
Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Case Party building-led practices in cost reduction efficiency enhancement and production capacity breakthroughs Construction of Corporate Governance System
Sanjing Mingshui focusing on key tasks such as cost control expense reduction and capacity enhancement The Company strictly complies with the Company Law of the People's Republic of China the Securities Law of the People's Repub-
systematically advances the "Party Branch Secretary Leads Projects" special initiative. The Party Committee aligned lic of China and relevant regulatory requirements of the China Securities Regulatory Commission (CSRC) and stock exchanges.with operational realities selects topics scientifically and initiates projects precisely. Branch secretaries serve as It continuously enhances its corporate governance structure and internal governance systems standardizes the operational
primary project leaders forming Party-member-led task forces and establishing mechanisms for planning progress mechanisms of authority and responsibility and ensures lawful and compliant operations. By strengthening the governance
tracking and scheduling to ensure orderly implementation. Projects target critical areas including API capacity framework comprising the Shareholders' Meeting the Board of Directors and its specialized committees and management level
expansion process optimization and energy system upgrades achieving substantive progress in increasing batch the Company promotes scientific decision-making standardized operations and steady development laying a solid governance
output optimizing production efficiency and stabilizing product quality. By leading breakthrough projects through foundation for its sustainability.Party building grassroots organizational vitality is further energized providing strong support for the Company's
achievement of annual business objectives. Shareholders' Meeting Audit Committee
Strategy and Sustainability Committee
Board of Directors
Remuneration and Appraisal Committee
President
Nomination Committee
Secretary Chief Vice Vice Vice Vice
of the Financial President of President of President of President of
Board of Officer R&D Production Marketing Operations
Sanjing Mingshui's "Party Branch Secretary Leads Projects" Initiative
Directors
Governance structure
Case Party building-led practices in technological breakthroughs and innovation
Sanchine focuses on technological innovation and quality improvement using the "Party Branch Secretary Leads
Projects" initiative as a key vehicle to advance Party-building empowerment. It channels the strength of Party organi-
zations toward the front lines of R&D and production. Each Party branch establishes task-force projects aligned with
key priorities such as production process optimization quality and cost control and automation upgrades and
organizes Party-member technical experts to advance them collaboratively. Through measures including equipment
parameter optimization process flow improvements and automation enhancements these projects have achieved
phased results in reducing production losses increasing finished product yield and advancing intelligent manufac-
turing. The project-based approach under Party leadership has effectively fostered synergistic alignment between
technological breakthroughs and business development.Management Group Photo
Sanchine's "Party Branch Secretary Leads Projects" Initiative Focusing on corporate governance and standardized operations the Company has established and continuously improved its
governance system and rules of procedure including the Rules of Procedure for Shareholders' Meetings the Rules of Procedure
for the Board of Directors the Independent Director System and working rules for specialized committees. These institutional
arrangements clearly define the boundaries of authority and responsibility and operational mechanisms of governance bodies
providing an institutional guarantee for the independent and effective performance of the Board of Directors.
25 26About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
The Company has established a governance structure characterized by clear
delineation of powers and responsibilities standardized operations and mutual During the reporting period the Company
checks and balances. The Shareholders' Meeting composed of all shareholders
serves as the Company's governing authority and exercises decision-making
power over major corporate matters in accordance with the law. Through Had independent directors Had female directors
standardized procedural rules it ensures that all shareholders can exercise their accounting for??.?% accounting for ??.?%
rights lawfully and equally. The Board of Directors is accountable to the Share-
holders' Meeting and fulfills its statutory decision-making and supervisory
duties. Senior management personnel are appointed by the Board of Directors
and are responsible for the Company's day-to-day business operations and Annual General Meeting The Board of Directors fulfills its statutory supervisory responsibilities regarding the Company's strategic planning business
implementation of Board resolutions. management and risk control. By convening board meetings regularly it reviews key matters related to the Company's business
development ensuring that Board operations are standardized orderly and efficient. This guarantees compliance with
To safeguard the independence and effectiveness of the Board of Directors the Company has implemented institutional arrange- decision-making procedures and truthful accurate and complete information disclosure thereby effectively safeguarding the
ments such as the Independent Director System and the Working System for Special Meetings of Independent Directors. These Company's overall interests and the legitimate rights of all shareholders.provide a mechanism to support independent directors in performing their duties lawfully independently and prudently thereby
enhancing the Board's oversight and checks-and-balances capacity in decision-making on major matters and improving the
standardization and effectiveness of its overall operations.During the reporting period the Company
Case Special research to strengthen support for independent directors in fulfilling their duties
Held??Board of at which??matters Achieved a director Organized ?director
In July 2025 the Board of Directors' Office of the Company organized a special research session for independent
Directors meetings were reviewed and approved attendance rate of???% training sessions
directors featuring thematic reports and discussions on GNC China's business operations marketing system
development strategy execution and cross-border business expansion. Drawing on their industry experience and
regulatory insights the independent directors provided targeted recommendations on business operations and
risk management. On-site engagement deepened their understanding of industrial chain operations and gover-
nance practices.The Company continues to strengthen independent To further enhance the professionalism and scientific basis of Board decision-making the Company's Board of Directors has
directors' familiarity with key business areas and operational established four specialized committees: the Audit Committee the Strategy and Sustainability Committee the Nomination
processes through special research joint business visits Committee and the Remuneration and Appraisal Committee. Each committee carries out its duties according to defined responsi-
performance-related training and information exchange bilities and provides support and recommendations to the Board on relevant professional matters thereby promoting scientifically
mechanisms enabling more informed and targeted profes- grounded decision-making and effective performance of duties.sional judgment in decision-making and oversight. These
arrangements have gradually evolved into a regularized
support mechanism for director duties effectively enhanc- During the reporting
period the Company During the reporting period the Company was awardeding information transparency and communication efficiency
and promoting more standardized stable and efficient
corporate governance.Held?Audit Committee
meetings
Board Diversity Held?Strategy and
Sustainability
The Company specifies in its Rules of Work for the Nomination Committee of the Board of Directors that in the nomination and Committee meeting
appointment of directors it comprehensively considers candidates' professional backgrounds industry experience performance
capabilities and independence. Diversity is treated as a key criterion to continuously optimize the composition of the Board of
Directors and foster a structure that integrates diverse perspectives and complementary expertise. Held?Nomination
Committee meeting
As of the end of the reporting period the Company's Board of Directors consisted of nine directors including one employee repre-
sentative director three independent directors and four female directors. The Board exhibits a relatively diverse composition in Held?Remuneration 2025 Excellent Practice for 2025 Excellent Practice Case 2025 Grade 5A Certificate
terms of gender functional roles and professional backgrounds which helps introduce varied perspectives into the decision-mak- the Board of Directors of a for the Board of Directors' for Secretary of the Board and Appraisal Listed Company Office of a Listed Company Performance Evaluation of
ing process thereby enhancing the quality of deliberations and the scientific rigor of decisions. Committee meetings Listed Companies
27 28About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
The Company consistently focuses on protecting the rights of small and medium-sized investors—including their right to informa-
Investor Relations Management tion right to participation and other legitimate interests. By creating diverse and accessible communication scenarios it advanc-es investor relations management from one-way information dissemination toward two-way interaction thereby deepening small
The Company places great importance on investor relations management recognizing it as a critical component for enhancing and medium-sized investors' understanding of and confidence in the Company's governance and long-term value.corporate governance transparency strengthening market recognition and protecting investors' legitimate rights and interests. It
has formulated and implemented the Investor Relations Management System and the Market Value Management System which
clearly define responsibilities workflows and communication standards for investor relations activities ensuring their lawful Case Special research to strengthen support for independent directors in fulfilling their duties
compliant and orderly execution.In September 2025 the Company participated in the "I am
a Shareholder" event for listed companies organized by
the Shanghai Stock Exchange. In collaboration with the
China Association for Public Companies the China
Transparent information disclosure Diversified communication channels
Securities Investor Services Center and the Shanghai
Securities News it invited more than 20 small and
medium-sized investors for an on-site visit and exchange.Aligning with key capital market concerns—such as To ensure the stability and effectiveness of During the event investors toured the Sanchine exhibi-
operating performance development strategy risk communication channels the Company has estab- tion center and the intelligent oral liquid production line
management and sustainability—the Company lished a diversified and regularized investor gaining direct insight into the Company's development
maintains ongoing communication with investors communication mechanism. Leveraging its official journey product portfolio and achievements in intelligent
and shareholders through multiple channels. It website the Shanghai Stock Exchange's "SSE manufacturing. Face-to-face discussions enabled a
provides timely updates on its operational status e-interaction" platform the investor hotline deeper understanding of the Company's strategic
business layout and development plans while earnings calls and Shareholders' Meetings it planning and operational status. The event also featured a
actively soliciting market feedback and construc- provides investors with reliable and convenient dedicated investor education session titled "Know Your "I am a Shareholder" visits Shanghai-listed company—
tive suggestions to enhance the transparency and access to corporate information and opportunities Hapharm GroupRights Exercise Your Rights Protect Your Rights" aimed
comprehensibility of its disclosures. for interaction. at enhancing small and medium-sized investors' aware-
ness of rights protection.During the reporting period the Company During the reporting period the Company was awarded
Disclosed ?periodic announcements and??ad-hoc announcements;
Organized? roadshows and held communications with a total of?? institutional investors;
Conducted?? reverse roadshows hosted ?? securities firms and institutional investors with a total Certificate of Excellent Practice for
the 2024 Annual Report Earnings
of???participants; Call of Listed Companies
Received and answered a total of??? investor inquiries via the investor hotline;
Received a total of???questions from investors with a response rate of??.??%
29 30About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability
Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation
to Build a Safe and
Management Shared Future
Strategy
Internal Control and Compliance Management Supported by its institutional framework the Company integrates compliance requirements throughout the entire cycle of
The Company values internal control and compliant operations continuously refining relevant systems and management mecha- decision-making execution and supervision extending them to all departments subsidiary enterprises and employees. This
nisms. It embeds compliance requirements into all aspects of business management steadily enhancing its level of standardized facilitates the transition of compliance management from system establishment to normalized operation. Additionally from the
operations and risk prevention capabilities to support sustainable and stable development. perspectives of operational stability and long-term development the Company systematically identifies compliance-related risks
and opportunities using these insights to continuously refine management measures and optimize resource allocation.Risk name1 Specific description Time horizon2 Financial impact Response measures
Compliant OperationIncreased costs Establish a “Three LinesThere are instances where compli- related to non-com- of Defense” compliance
Governance Compliance ance requirements are not fully Medium and pliance penalty management system and
management
implemented during their embedding long term expenses and strengthen enforcement
The Company regards lawful and compliant operations as a fundamental requirement for business management and employee execution risk and execution in business processes potential litigation of policies and supervi-
conduct. Building upon national laws and regulations it has established a compliance management system centered on the losses sory inspections
Compliance Management Measures of HARBIN PHARMACEUTICAL GROUP CO. LTD. supplemented by specialized policies and
operational guidelines. During the reporting period the Company issued three new guidelines: Compliance Guidelines for Prevent- Elevated risk of Regularly conduct n key areas such as fund security tax
ing Commercial Bribery in the Pharmaceutical Sector Advertising Compliance Guidelines and Compliance Guidelines for fund loss higher compliance risk Compliance handling trade secrets and personal
Livestreaming Marketing Activities further strengthening institutional controls over key risks in the pharmaceutical field. compliance costs identification and risks in key information protection risk identifica- Medium-term
and increased assessment and
areas tion is insufficient or controls are
potential compen- formulate and implement
The Company has established a compliance management organizational system centered on the Compliance Committee and the inadequately implemented sation expenses risk response plans
Chief Compliance Officer supported by a management mechanism featuring clearly defined responsibilities and coordinated
operations. It has implemented a compliance management framework based on the "three lines of defense" embedding compli- Opportunity name Specific description Time horizon Financial impact Response measures
ance responsibilities across management levels and business processes to ensure effective implementation of compliance
requirements throughout the organization. Embed compliance The compliance management system
requirements into the
Opportunity for and processes are continuously Reduced compli-
entire process of
management improved and the integration of Medium and ance costs and
Compliance Committee decision-making execu-efficiency compliance requirements into long term decreased
tion and supervision to
improvement business processes is progressively operational losses
Responsible for the organizational leadership and overall coordination of compliance promote process standard-enhanced
management holding regular meetings to study and decide on major compliance matters ization
or propose recommendations and to guide supervise and evaluate compliance manage- Compliance risk identification and Continuously carry out risk
ment activities. Reduced risk-relat-Risk and cost proactive management mechanisms identification classified
Medium and ed losses and
control are gradually strengthened and risk management and the
long term improved cash flow
opportunities control capabilities are continuously development of response
stability
enhanced plans
First line of defense Second line of defense Third line of defense
Discipline inspection and supervision
All departments of the Compliance Management Case Building a compliance assurance system to promote full-staff compliance practices
agencies and audit supervision and
Company Department
accountability departments Centered on the principle of "Compliance Escorting High-Quality Develop-
ment" the Company has established a "1-2-3-10+" compliance assurance
Bear direct and primary responsi- Responsible for organizing Within their scope of authority
system anchored by the Compliance Management Measures and the Employ-
bility for compliance management coordinating guiding and supervis- they supervise the implementation
ee Compliance Manual. It is supported by three checklists—covering compli-
within their scope of duties and are ing compliance management of compliance requirements
ance obligations/risks responsibilities of key positions and key process
responsible for daily compliance activities and providing compliance investigate violations and carry out
controls—and guidelines for priority areas such as anti-commercial bribery in
management in their respective support to other departments accountability procedures as
the pharmaceutical sector and advertising compliance effectively embedding
business areas or departments. (subsidiary enterprises). stipulated.compliance requirements into business processes.Through mechanisms such as compliance reviews risk early-warning systems
Compliance Administrator Compliance Management Specialist Compliance Administrator and violation reporting channels the Company achieves comprehensive cover- "1-2-3-10+" compliance assurance
age across its 18 subsidiary enterprises transitioning compliance from institu- system
tional design to daily execution and employee practice. This provides
Compliance administrators receive business guidance and training from the compliance management
sustained assurance for stable operations.department and provide compliance advice to their department heads for decision-making reference.Organizational structure of Hapharm Group compliance management 1 The risks and opportunities listed in this table are forward-looking identifications made by the Company based on changes in external environment and
internal operational conditions. They are intended to reflect the potential impact trends and key management priorities and shall not constitute a definitive
judgment on future events nor indicate that relevant risks have materialized or exerted significant impacts on the Company’s current operations.
2 Impact time refers to the timeframe over which a significant impact is exerted on the Company: short-term—within 1 year; medium-term—1 to 5 years;
31 long-term—over 5 years. 32About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Focusing on the compliance management goals of "comprehensive coverage efficient operation controllable risks and cultural Impact risk and opportunity management
consciousness" the Company continuously improves its compliance management system supported by organization systems
operations and assurance embedding compliance requirements into the entire process of business management and employee Focusing on the goal of lawful and compliant operations the Company embeds the identification and management of compli-
performance. By compiling and issuing the Employee Compliance Manual and organizing all employees to sign the Compliance ance-related impacts risks and opportunities into the entire process of business management. It achieves unified management
Commitment Letter it clarifies employee compliance obligations and behavioral boundaries. through system development process control and supervisory collaboration. Relying on the Compliance Committee the Chief
The Company upholds the compliance culture philosophy of Compliance Officer and relevant functional departments and in conjunction with actual business operations it continuously
"acting within principles and rules; with reverence and identifies potential compliance risks and their potential impacts in key areas such as fund security tax management trade secret
self-restraint" and promotes the building of a compliance protection and personal information protection. It then conducts a graded assessment based on the likelihood and impact of
culture through a combination of training and advocacy. these risks. For the identified key risks the Company clarifies management responsibilities through a "three lines of defense"
Relying on its annual compliance training plan it conducts ??? mechanism promoting the effective implementation of risk response measures in system execution daily supervision and rectifi-% signing rate for the
compliance education for key positions and new employees Compliance Commitment Letter cation. Through continuous tracking and dynamic assessment it constantly optimizes its compliance management system releas-
covering topics such as anti-commercial bribery business ing potential opportunities such as improved management efficiency and risk cost control while preventing risks.compliance and professional conduct. Through diversified
advocacy formats it strengthens warning and guidance Indicators and objectives
continuously enhancing employees' compliance awareness.The Company takes enhancing the effectiveness of compliance management and the level of compliance risk prevention and
control as the core goal of its compliance management. By improving the compliance management system it reduces the proba-
bility of major compliance risks ensures that business activities are conducted in accordance with laws and regulations and
Case Building legal and compliance capabilities in key business areas safeguards its operational stability and long-term development foundation.In June 2025 the Company organized a special topic lecture on business legal compliance focusing on key Indicator name Unit ????
business areas such as distributor management and live-streaming operations. An external team of professional
lawyers was invited to deliver the lecture. The training focused on distribution and agency contract management Number of compliance risks identified during the year Items 156
rebate and third-party expense control legal risks in online live-streaming promotion and vertical monopoly
compliance requirements. A total of 249 employees participated online and offline. Through special topic explana- Number of compliance risk response plans formulated Copies 15
tions and case analyses participants further identified compliance risk points in their business operations and
Number of compliance training sessions conducted Times 5
enhanced their contract management and risk prevention capabilities.Number of employees covered by compliance training Person-times 1010
Internal Control
The Company continuously improves its internal control management system regarding internal control as an important founda-
tion for standardizing business management preventing operational risks and enhancing governance effectiveness. Under the
supervision and guidance of the Board of Directors and its Audit Committee management coordinates and promotes work related
to internal control and internal audit pushing for internal control requirements to cover the entire process of decision-making
execution and supervision and ensuring their implementation throughout the Company and its subsidiaries.The Company has established and operates an internal control evaluation mechanism where management organizes and
conducts self-assessments of internal control to systematically evaluate the design and execution effectiveness of its systems.Combined with the results of internal supervision and external independent audits it continuously examines the operation of
internal controls.The Company formulates an annual internal audit plan to audit the business management and internal control operations of the
Company and its subsidiaries. It also promotes the rectification and implementation of issues found during audits forming a
closed-loop management system. During the reporting period the Company conducted 19 internal audit projects covering key
business processes such as procurement marketing fund usage engineering projects asset management financial accounting
budget execution and contract management. No major issues were found.Compliance Training Session
33 34About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to
from the Hapharm Performance in Special Topic to Solidify a Foundation for Product System for Value
Employee Development
Report Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Culture of Integrity and Honest Operations Anti-Unfair Competition
The Company adheres to lawful compliant and honest operations making integrity building an important part of its corporate Focusing on anti-monopoly and anti-unfair competition management the Company strictly implements the requirements of laws
governance and compliance management. It continuously improves its working mechanisms for anti-commercial bribery anti-cor- and regulations such as the Anti-Monopoly Law of the People's Republic of China and the Anti-Unfair Competition Law of the
ruption anti-monopoly and anti-unfair competition as well as for building an integrity culture and protecting whistleblowers. It People's Republic of China. In conjunction with the Company's and the sector's business characteristics it has formulated and
promotes the integration of integrity requirements into its business management and the entire process of employee perfor- implemented the Anti-Monopoly Management Regulations integrating the principle of fair competition into its business manage-
mance fostering a fair transparent and standardized business environment. ment and employee performance standards. The Company refines and adopts relevant legal requirements through institutionalized
means clarifies internal management requirements and promotes the enforceability and operability of anti-monopoly and
anti-unfair competition requirements within the Company.Anti-Commercial Bribery and Anti-Corruption
Focusing on anti-commercial bribery and anti-corruption management the Company strictly implements the requirements of Whistleblower Protection
relevant laws and regulations such as the Criminal Law of the People's Republic of China and the Anti-Unfair Competition Law of
the People's Republic of China. In conjunction with its actual business management it has formulated and implemented the The Company attaches great importance to the protection of the legitimate rights and interests of whistleblowers making
Anti-Commercial Bribery Management Measures which clarify the behavioral boundaries for employees in business dealings and whistleblower protection an important component of its integrity building and internal supervision mechanism. The Company has
in the course of their duties and integrate integrity requirements into the Company's business management and employee perfor- clarified confidentiality requirements for reported information at the institutional level and strictly implements the Provisions on
mance standards. Confidentiality in Case Handling by the Discipline Inspection Commission of Harbin Pharmaceutical Group Holding Co. Ltd1. It
rigorously manages whistleblowing clues the identity of whistleblowers and related information to prevent leaks or improper use.Reporting Mechanism Integrity Commitment Letter he Company prohibits any form of retaliation and promptly addresses and handles any potential adverse effects arising from
The Company has established and operates a reporting mecha- In accordance with the unified arrangements of the Group’s whistleblowing activities. It strives to create a favorable environment where supervision is encouraged and conducted in accor-
nism for commercial bribery and corruption encouraging Discipline Inspection Commission the Company organized dance with the law providing institutional guarantees for integrity building.employees and relevant parties to report violation clues in all management personnel to sign the Commitment to
accordance with laws and regulations. It has also set up Integrity in Practice achieving a 100% signing rate. For
dedicated channels such as a telephone line and an email confirmed cases of commercial bribery and corruption the Integrity Culture Building
address to receive reports related to commercial bribery and Company conducts investigations and handles them in
corruption. accordance with the law and regulations based on relevant
provisions such as the Regulations on Disciplinary Actions The building of a culture of integrity continues to be synergistically advanced with the building of corporate culture. Through a
Reporting hotline:????????-????? by the Communist Party of the People's Republic of China. It combination of institutional guidance publicity and education and practical activities the Company guides the integration of
holds the relevant parties seriously accountable to maintain integrity requirements into employees' daily work and codes of conduct.Reporting email: hyjtjw@???.com a standardized and orderly business environment.Relying on platforms such as the discipline enforcement and supervision section of its WeChat official account bulletin
During the reporting period the Company boards and electronic screens the Company conducts regular publicity on integrity and compliance. It sets up special
topics such as interpretations of Party discipline and regulations and building of an integrity culture gradually forming a
stable platform for disseminating the culture of integrity. During the reporting period a total of 86 related articles were
published with nearly 10000 views. In conjunction with actual business management and the characteristics of key
positions the Company conducts multi-level warning education. Through visits to integrity education bases holding
warning education meetings and using cases to explain discipline and law it guides employees to be reverent and adhere
to the bottom line. During the reporting period Party organizations and discipline inspection agencies at all levels
organized more than 90 warning education activities covering over 6300 participants. At the same time through activi-
ties such as integrity-themed essay contests short video creation and integrity-focused home visits the culture of
??? ?? ???? integrity is extended to employees' work and life scenarios promoting the transformation of integrity concepts from Achieved % Conducted Organized
institutional constraints to inherent self-discipline and creating a favorable environment for stable operations.integrity risk assessment anti-commercial bribery person-times of anti-com-
coverage for the Company and anti-corruption mercial bribery and
and its subsidiaries training sessions anti-corruption training for
its employees
1 The Company’s discipline inspection supervision and Party committee-related functions are centrally managed by or implemented with reference to
the Group without affecting the effective operation of the Company’s governance structure and internal oversight mechanisms.
353602
Product System
for Value Creation
The content of this chapter
Technological R&D and Innovation Practices ??
Indicators and Objectives ??
Industrial Cooperation and Exchange ??
Product Quality and Safety Management ??
Customer Rights and Interests and Service Assurance ??
Supply Chain Management and Security ??
Information Security and Privacy Protection ??
Medication Accessibility Enhancement ??About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon Creation to Build a Safe and
Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Management Shared Future
Opportunity name Specific description Time horizon Financial impact Response measures
Technological R&D and Innovation-Driven Development Continuously to shift its
R&D model from "imita-
tion-innovation integra-
Governance Against the backdrop of innova- tion imitation-oriented" to Development Increased revenue
tion-driven pharmaceutical develop- "imitation-innovation
The Company continuously optimizes its R&D management system implementing unified management and hierarchical advance- opportunities for Medium and higher gross margin ment the company continues to integration innovation-ori-
innovative drugs and long term and enhanced asset
ment for R&D activities and has formed an R&D organizational structure of "one department and five institutes" to carry out this advance its R&D portfolio of innova- ented" and strengthens
improved new drugs value
approach. tive drugs and improved new drugs project initiation and R&D
investment in innovative
The Company has established a Scientific Research Management Department responsible for the overall coordination organiza- drugs and improved new
tion and management of R&D work. Under its unified management five specialized research institutes have been set up: the drugs.Hapharm Drug Research Institute Hapharm New Drug Research Institute Hapharm Wellness Research Institute Hapharm Biologi-
Promote the systematic
cal Research Institute and Hapharm Traditional Chinese Medicine Research Institute. These institutes respectively carry out R&D The secondary development of research and secondary
work in the fields of generic drugs innovative drugs OTC and health products biological products and traditional Chinese Opportunities for the established traditional Chinese development of
medicine. By clarifying management levels and responsibilities the Company achieves integrated allocation and specialized secondary develop- Enhanced value of medicine (TCM) products is ongoing established traditional
collaboration of R&D resources promoting the orderly conduct of R&D activities within a unified management framework. ment of established Medium-term existing assets and continuously extending product life Chinese medicine
traditional Chinese increased revenue
cycles and expanding application products and continuous-
medicine products
value ly optimize the product
Strategy pipeline structure
In accordance with overall development planning and R&D strategy the Compa-
ny adheres to innovation orientation. With the core goals of enriching and Construction of the R&D System
building product echelons and optimizing R&D system and team it continues to The Company focuses on five major disease areas: cardiovascular and cerebrovascular digestive and metabolic respiratory
transform the R&D model from "imitation-innovation integration imitation-ori- anti-infective and anti-tumor as well as three key directions: geriatric medicine pediatric medicine and health products. It has
ented" to "imitation-innovation integration innovation-oriented". Focusing on built an product system featuring the coordinated advancement of chemical drugs traditional Chinese medicine and biological
key R&D fields the Company keeps improving its R&D and technological innova- drugs supplemented by differentiated health products. The R&D project initiation focuses on generic drugs with high technical
tion system and implements its five-year R&D strategy and action plan. With the barriers and innovative drugs while simultaneously promoting the secondary development of established traditional Chinese
vision of building a century-old enterprise with sustainable sound development medicine varieties with clinical value continuously optimizing the product pipeline structure and cultivating a competitive product
and becoming a benchmark in China’s pharmaceutical and health industry the echelon.Company establishes long-term R&D thinking making R&D an important
In terms of platform and capability building the Company continuously
foundation supporting its high-quality development. During the implementation
improves its technology platform system adapted to its own R&D charac-
of R&D and innovation strategy the Company identifies and analyzes major risks During the reporting period
teristics. It introduces key technological paths such as continuous flow
and potential opportunities related to R&D and takes corresponding counter- R&D Strategy Promotion Conference biosynthesis peptide synthesis taste-masking technology and sustained
measures.and controlled release to enhance R&D efficiency and product differentia- Shiyitang Traditional Chinese Medicine
tion. At the same time it regards the cultivation of scientific research has been successively accredited as
Risk name Specific description Time horizon Financial impact Response measures talent as an important support for the R&D system. Through a combina- municipal and provincial Postdoctoral
tion of project practice platform construction external training and Innovation Practice Base.Rationally allocate R&D academic exchanges it promotes R&D-production synergy and technolo-
Increased R&D invest-
The new drug R&D process involves a resources advance R&D gy inheritance solidifying the long-term foundation for R&D development.Risks related to the ment and greater
long development cycle technical Medium and projects in stages and
new drug R&D cycle uncertainty in the
complexity and uncertainty in interim long term strengthen dynamic
and uncertainty recovery of capitalized management of R&D Case
Jointly building the Cold Region Traditional Chinese Medicine R&D Platform to solidify the foundation of
outcomes industrial innovationexpenditures
projects
To advance the resource protection and modernization of Chinese materia medica in cold regions Shiyitang Chinese
Medicine and Northeast Forestry University signed the Letter of Intent for the Construction of a Joint R&D Center for
Strengthen the evaluation Cold-Region Longjiang Medicines to jointly build the "Joint R&D Center for Cold-Region Longjiang Medicines" and
Risk of intensified of R&D project initiation
In the high-barrier generic drug on this basis jointly establish a postdoctoral research workstation.competition for Declining revenue and focus on selecting
segment market competition is
generic drugs with Medium-term compressed profit varieties with technical intensifying and pressure on R&D and
high technical margins barriers and enhance R&D The two parties will cooperate in areas such as resource protection of authentic Chinese materia medica in cold
regulatory registration is increasing
barriers efficiency and product regions GAP standardized planting construction of a quality traceability system and research on new functions and
differentiation components of traditional Chinese medicine. The initial cooperation period is planned for 2025–2029 aiming to
promote the transformation of scientific research achievements and the cultivation of professional talent thereby
boosting the innovation capabilities and scientific research level of the regional traditional Chinese medicine industry.
39 40About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Hapharm Performance in Special Topic to Solidify a Foundation for Product System for Value
Employee Development
Report from the Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Green R&D and Green Products
Case Deepening industry-academia-research cooperation to promote the building of TCM innovation capabili-ties
In 2025 focusing on the innovative development of traditional Chinese The Company integrates green concepts into the entire R&D process practicing the R&D policy of "green R&D + safety and
medicine Shiyitang Chinese Medicine organized its R&D team to conduct quality." Focusing on reducing energy consumption minimizing pollution and enhancing resource utilization efficiency it continu-
exchanges and investigations with universities including Harbin Institute ously promotes the application of green technologies in the R&D of pharmaceuticals and health products. Through technological
of Technology Beijing University of Chinese Medicine Heilongjiang paths such as synthetic biology and continuous flow the Company optimizes process flows at the R&D stage reducing raw materi-
University of Chinese Medicine and scientific research institutions such al consumption and environmental load while improving reaction efficiency and product stability.as Tianjin Pharmaceutical Research Institute. Special seminars were held
on innovative TCM drugs improved TCM drugs evidence-based medicine In specific practice synthetic biology technology centered on biological systems achieves sustainable raw material conversion
research and post-marketing evaluation of TCM injections. and highly selective synthesis; continuous flow technology centered on process intensification enhances the safety stability and
resource utilization of reaction processes. These green technologies play a supporting role in the R&D and process design stages
Through sustained industry-university-research exchanges the Company laying the foundation for the industrial application of green processes and driving product R&D towards low energy consumption
reached cooperation consensus on multiple research fields and promoted low emissions and high quality.the signing of letters of intent. This laid a foundation for subsequent R&D
Exchange Visit with Tianjin Pharmaceutical Delivery Unit Mixing Unit Reaction Unit Back Pressurecooperation and achievement transformation and further improved the Research Institute Unit
enterprise’s innovation capacity and R&D system construction.Reagent
A
Product
The Company simultaneously improves its incentive and project management mechanisms for R&D personnel. It has formulated
and implemented the R&D Project Incentive Plan of HARBIN PHARMACEUTICAL GROUP CO. LTD. (Trial). Centered on the full Reagent
life-cycle management of R&D projects this plan links project progress with the performance of R&D teams by setting quantitative B
assessment indicators at key milestone nodes. This forms an achievement-oriented incentive mechanism enhancing the motiva- Continuous Flow Schematic Diagram
tion of R&D personnel and the efficiency of project execution and providing a dynamic guarantee for the continuous advancement
of the R&D strategy. Intellectual Property Protection
The Company continues to increase its R&D investment supporting the implementation of its R&D strategy with an institutional-
ized investment guarantee mechanism. It advances projects through a combination of independent R&D and external cooperation. The Company conducts intellectual property management in accordance with the law strictly adhering to the requirements of laws
During the reporting period over a hundred projects were initiated covering new drugs generic drugs and biological products and regulations such as the Civil Code of the People's Republic of China the Trademark Law of the People's Republic of China
with more than 50 projects having been approved or completed. The Company and its subsidiaries have obtained multiple national and the Patent Law of the People's Republic of China to implement full-process management of trademarks patents and
provincial and municipal innovation qualifications and have been recognized as scientific research platforms providing support copyrights. The Company has formulated and implemented the Regulations on the Management of Patents and Monographs to
for the implementation of the R&D strategy and the transformation of its achievements. standardize matters such as application maintenance rights management and termination. In terms of trademark management
it has implemented the Regulations on the Management of Trademarks of HARBIN PHARMACEUTICAL GROUP CO. LTD. clarifying
the requirements for trademark registration use licensing and maintenance to prevent infringement and improper use risks.Certifications/honors
On the basis of system implementation the Company enhances its intellectual property protection capabilities through training
National High-tech Enterprise and exchanges. It conducts training for R&D personnel new employees and relevant positions on topics such as patent basics
HARBIN PHARMACEUTICAL GROUP CO. LTD. National Intellectual Property Advantage Enterprise innovation point mining rights protection practices and standardized trademark use. It also organizes participation in sector
Harbin Pharmaceutical Group Sanjing Qianhe Pharmaceu- (Demonstration) intellectual property exchange activities to continuously strengthen employees' compliance awareness and professional capabili-
tical Co. Ltd. Harbin Pharmaceutical Group Bioengineering Co. Ltd. ties promoting the effective protection and transformation of innovative achievements.Harbin Pharm Group Sanjing Pharmaceutical Co. Ltd. Heilongjiang Province "Specialized Refined Differential
Harbin Pharmaceutical Group Bioengineering Co. Ltd. and Innovative" Small and Medium-Sized Enterprise Impact Risks and Opportunities
Harbin Pharmaceutical Group Sanjing Mingshui Pharma-
ceutical Co. Ltd. Harbin Pharmaceutical Group Bioengineering Co. Ltd.Harbin Shiyitang Chinese Medicine Co. Ltd. Harbin Shiyitang Chinese Medicine Co. Ltd. In R&D activities the Company simultaneously identifies operational impacts risks and development opportunities brought by
Harbin Pharmaceutical Group Sanjing Daqing Glass Heilongjiang Province Technological Innovation Demon- technological innovation and integrates relevant management requirements into R&D decision-making and project management
Industrial Park Co. Ltd. stration Enterprise processes. At key links including R&D project initiation project evaluation and annual planning the Company comprehensively
Harbin Pharmaceutical Group Shiyitang Chinese Decoction considers technical feasibility market demand policy environment and resource allocation. It systematically identifies and assess-
Harbin Pharmaceutical Group Bioengineering Co. Ltd.Pieces Co. Ltd. es the uncertainty of cost cycle and achievement in R&D activities.National Torch Program Harbin Antibiotic Heilongjiang Provincial Enterprise Technology Center Via milestone management phased evaluation and dynamic adjustment mechanisms the Company maintains continuous supervi-
Specialty Industrial Base Harbin Pharmaceutical Group Bioengineering Co. Ltd. sion over R&D projects and optimizes R&D pipeline and resource allocation based on evaluation results. While preventing R&D
HARBIN PHARMACEUTICAL GROUP CO. LTD. HARBIN PHARMACEUTICAL GROUP CO. LTD. risks and improving resource efficiency it promotes the transformation of technological innovation achievements and sustains
the supporting effect of innovation on core competitiveness and long-term development.
41 42About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth
Creation Management Shared Future
Indicators and Objectives Industrial Cooperation and Exchange
Focusing on R&D and innovation work the Company has defined its core management objective as improving the operational
Grounded in the broader context of the pharmaceutical and health industry's development the Company actively integrates into
quality of the R&D system and the efficiency of achievement transformation. By strengthening the overall planning of R&D project
the collaborative development patterns of the industrial and innovation chains. Focusing on key directions such as the inheritance
screening and process management it continuously optimizes the structure of the R&D pipeline and the efficiency of resource
and innovation of traditional Chinese medicine the enhancement of fundamental industrial capabilities and the transformation of
allocation. The Company focuses on strengthening the layout and management of projects with high technical barriers and high
scientific and technological achievements it continuously expands the depth and breadth of industrial cooperation. This promotes
clinical value promoting a positive cycle between R&D investment and product outcomes and business benefits thereby provid-
the effective connection of industry academia research and application contributing to the overall improvement of the industrial
ing sustained support for the Company's long-term and steady development.chain and the high-quality development of the regional pharmaceutical industry.Indicator name Unit 2025
Case Participating in the industrial collaboration of Chinese materia medica in cold regions to promote the Total R&D investment Ten Thousand Yuan 20351.82 development of regional industrial chains
R&D Expenditure as a Percentage of Operating Revenue % 1.27 In March 2025 Shiyitang Chinese Medicine as one of the
R&D Expenditure as a Percentage of Operating chain-leader enterprises in Heilongjiang province's Chinese
Revenue of Industrial Sector % 3.54 materia medica industry chain co-organized the "Cold-Region
Proportion of R&D personnel % 10.54 Longjiang Medicines · Acanthopanax Senticosus" Industry Devel-
opment Conference. The conference focused on the resource
Total Amount of Science and Technology
Innovation Awards Issued Ten Thousand Yuan 215.33 protection standardized cultivation industrial synergy and value
Number of Recognized Science and Technology enhancement of authentic Chinese materia medica in cold regions
Innovation Projects Items 146 like Acanthopanax senticosus. It brought together representatives
Total number of valid patents held Items 220 from relevant provincial government departments scientific
research institutions industry associations and enterprises for
Total number of invention patents applied for in the year Items 24 exchanges and discussions to jointly explore collaborative devel-
Total number of invention patents granted in the year Items 1 opment paths for the Chinese materia medica industry chain. By
building a multi-party communication platform the Company The First IP Image of Hailong Chinese Herbal Medicine –
Total number of valid trademarks held Items 774 actively promotes the integration of resources and Shiyitang Vitality Xiaowu
upstream-downstream collaboration in the Chinese materia
medica industry fostering the construction and high-quality
development of a regional traditional Chinese medicine industrial
Ethics of Science and Technology cluster.The Company's business management revolves around pharmaceutical R&D technology development and related scientific and
technological activities with its business covering areas such as chemical drugs traditional Chinese medicine biological
products and health products. In the management of its scientific and technological activities the Company adheres to the princi-
ple of legal compliance making ethics of science and technology requirements an important prerequisite for R&D management
and scientific research cooperation to ensure that related activities are carried out in an orderly manner within a standardized
framework.The Company complies with national requirements related to technology ethics and research management such as Good Clinical
Practice the Measures for the Ethical Review of Life Sciences and Medical Research Involving Human Participants and the
Measures for the Review of Ethics of Science and Technology (Trial). Through internal systems like the R&D Project Management
Measures it standardizes the initiation implementation and collaborative research of R&D projects. The systems stipulate that if
an R&D project involves clinical trials non-clinical studies or entrusts third parties to conduct related research activities it must
be executed in strict accordance with current national laws regulations and regulatory requirements. Compliance responsibilities
must also be clearly defined in cooperation or entrustment agreements to ensure that the entire research process meets ethical
and compliance requirements.The Company itself does not conduct research activities involving human clinical trials or animal experiments. In cases of commis- “Cold-Region Longjiang Medicines · Acanthopanax Senticosus” Industry Development Conference
sioned research or external cooperation the Company requires its partners to possess the corresponding qualifications by law and
imposes management constraints on their research conduct to ensure compliance with national ethics of science and technology
and regulatory requirements thereby preventing potential ethical and compliance risks at the source. During the reporting period
the Company had no instances of violating laws and regulations related to the ethics of science and technology.
43 44About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon
Creation to Build a Safe and
Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Management Shared Future
Product Quality and Safety Management Opportunity name Specific description Time horizon Financial impact Response measures
Continuously optimize
Product quality consistency and
Revenue growth the quality management
Governance stability are continuously Quality improvement Medium and enhanced brand system and strengthen improved and quality management
opportunity long term premium and quality control and
capabilities are steadily strength-
improved cash flow quality review through-
The Company always places product quality and drug safety at the core of its business management. Adhering to the principle of ened out the entire process
"compliance first full-process control and continuous improvement" it strictly follows the requirements of laws and regulations
such as the Drug Administration Law of the People's Republic of China Good Manufacturing Practice (GMP) and Good Supply
Practice for Drugs (GSP). The Company has established an integrated quality management system that covers the entire industri-
al chain and complies with industry regulatory standards. It has formulated and implemented policy documents such as the
Management System of the Comprehensive Quality Management Committee and the Quality Document Management Procedures
of the Joint-Stock Company to clarify quality management responsibilities processes and supervision mechanisms building a Full-Lifecycle Quality Management
quality management system that meets the regulatory requirements of the pharmaceutical sector.The Company systematically incorporates product quality and drug safety into its business management and quality governance
The Company continuously improves its quality management system structure. The Quality and Manufacturing Technology
systems gradually forming a management model guided by full-lifecycle quality management. The Company integrates quality
Department leads the overall coordination of quality management system development operational supervision and continuous
management requirements into every stage of the product life cycle from R&D to post-market surveillance. By combining system
improvement efforts. Each production operation and R&D entity in line with its own business characteristics strictly implements
development with process control it promotes the transformation of quality management from a singular focus on production
mandatory standards such as GMP and GSP. They formulate quality management system documents that match their product
control to a comprehensive front-end management approach providing continuous assurance for product quality stability and
types and business scopes ensuring seamless integration of quality management in organizational structure system execution
drug safety.and daily operations thereby comprehensively strengthening the foundation of quality control.? Product R&D stage ?Production and distribution stage ? Product post-market stage
Strategy
Adhering to the philosophy of The Company continuously The Company has established
The Company focuses its core efforts on building a compliant and efficient quality management system pursuing the "dual zero" "Quality by Design" the improves its quality manage- a mechanism for monitoring
goal and achieving quality excellence. It systematically enhances its quality management capabilities by focusing on areas such Company systematically ment mechanism covering raw adverse drug reactions and
as quality operation evaluation quality risk prevention and control digital and intelligent application in quality and the develop- conducts quality risk assess- and auxiliary material access quality feedback collecting
ment of professional talent echelons. Risks and opportunities related to product quality and safety are integrated into the daily ments process validations supplier audits production safety information through
operation and evaluation of the quality management system. and stability studies during process control finished multiple channels and
drug formulation design product inspection and release conducting analysis evalua-
Risk name Specific description Time horizon Financial impact Response measures process development and warehousing and transporta- tion and reporting in accor-
technology transfer. It defines tion and product traceability. It dance with regulatory
Strengthen standardized
product quality standards strengthens quality control of requirements. Concurrently
control throughout the entire
life cycle and process critical quality attributes and raw and auxiliary materials by the Company has formulated
enhance the precision of critical process parameters to implementing tiered audits and and implemented product
process control reinforce ensure that R&D outcomes dynamic management of recall management
Quality control measures in R&D quality awareness and have stable and reproducible suppliers. During the produc- documents which specify
Product recall losses
production or testing are professional capability production conditions before tion process it reinforces recall classification standards
Risk of quality Medium and compensation
inadequately implemented and the cultivation promote a entering the industrialization standardized requirements for disposal procedures and
control failure long term expenses and
quality management system has proactive risk prevention and
reduced revenue stage. Relevant R&D data and key processes critical responsibilities. When
operational weaknesses control mechanism and change information are equipment and operating potential quality or safety risks
establish a diversified quality managed and controlled in a personnel to ensure that are identified the Company
audit and supervision system standardized and traceable production activities remain can promptly initiate recall
to comprehensively build a
manner reducing the controlled. In the inspection measures of the appropriate
line of defense for quality
and safety likelihood of quality fluctua- and release stage an indepen- level leveraging its product
tions in subsequent produc- dent quality management traceability system to ensure
Continuously track tion and distribution stages function implements a that risks are controllable and
Drug regulatory policies and
Rising compliance regulatory changes improve from the source. multi-level review and release disposal is effective.technical standards are continu-
Regulatory compli- Medium and costs and heightened the quality management process and no product is
ously updated posing a risk of
ance risk long term risk of asset system conduct compli- allowed to enter the market
delayed alignment in the quality
impairment ance training and perform without approval.management system
regular system assessments
45 46About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability ReportCreation
Chairman Group Sustainability Stable Growth Management Shared Future
Based on the actual needs of product quality management the Company formulates an annual quality improvement plan defining
key improvement directions and implementation milestones and organizes its affiliated enterprises to advance quality improve- Case Honing elite skills through practice and demonstrating true expertise in competition
ment work as planned. Through regular internal quality audits management reviews and evaluations of improvement effective-
ness combined with dynamic monitoring and systematic optimization of the entire process the Company continuously enhances
the maturity of its quality management. This drives a steady improvement in product quality and drug safety levels effectively
safeguarding public safety and efficacy in medication use. In November 2025 the Company organized a "Qual-
ity Inspector Competition". The competition adopt-
ed a format combining "theoretical assessment and
Quality Management System Capabilities Building on-site practical operation" to comprehensively
evaluate the inspectors' understanding of regula-
tions professional judgment and operational
The effective operation of the quality management system relies on the combined efforts of system execution personnel capabili- standards. The theoretical assessment focused on
ties and organizational synergy. The Company continuously improves its quality management systems and business processes quality standards inspection principles and
systematically integrating quality management requirements into R&D production sales service and related business segments. problem analysis skills while the on-site practical
This promotes stable and effective linkage and operation of quality management across all stages. operation emphasized the execution of standard
operating procedures and actual inspection skills.To reinforce the implementation of quality responsibilities the Company uses positive incentive mechanisms to guide employees Through this competition which promoted learning
to proactively fulfill their quality duties. For key positions such as quality management production operations and inspection and and practice a platform for technical exchange in
testing it implements professional skill competitions job incentives and rewards for quality improvement suggestions. It also quality inspection was established. It effectively
links quality performance with individual evaluations and performance results ensuring that employees continuously improve enhanced the professional capabilities and regula-
their operational standardization and work quality in their daily work. tory awareness of the inspection personnel provid- Quality Inspection Staff Skill Contest
ing talent support for the stable operation of the
At the same time the Company regards professional capability building as a crucial guarantee for the long-term effective opera- quality management system.tion of its quality management system. Through a combination of systematic training and practical experience it continuously
enhances the understanding and execution capabilities of quality management production and inspection personnel regarding
regulatory requirements technical standards and quality risks. This approach promotes the transformation of quality manage-
ment from a system requirement to a level of excellence solidifying the foundation for the sustained and effective operation of
product quality and safety management.Case Heilongjiang Provincial Drug Quality Inspector Employee Professional Skills Competition
Case Enhancing professional capabilities in the quality technology sequence
In December 2025 the Company selected 13 contestants
The Company continues to advance the professional capability to participate in the Heilongjiang Province Drug Quality
building of its quality technology sequence. It has organized eight Inspector Professional Skills Competition with nine of
subsidiaries and affiliates to complete the compilation of the Enter- them achieving outstanding results including sweeping
prise-Level Quality Technology Professional Capability Manual and the first and second prizes. During the preparation
its supporting question bank. In accordance with the latest manage- period the Company determined the final contestants
ment requirements of the 2025 edition of the Pharmacopoeia of the through multiple rounds of competitive assessments and
People's Republic of China the Company has systematically invited professional instructors to conduct specialized
optimized the Quality Inspection Professional Capability Enhance- practical training to help them master key assessment
ment Manual of the Company. On this basis the Company simultane- points and enhance their competitiveness. The competi-
ously conducted specialized training and assessments for quality tion consisted of two parts: theory and practical opera-
technology inspection personnel covering eight enterprises to tion comprehensively testing the contestants' profes-
solidify the professional capabilities of the quality management sional knowledge and practical skills. Leveraging their
system. To promote and implement the new edition of the Pharmaco- solid foundation and craftsmanship the Hapharm
poeia of the People's Republic of China a total of 104 internal and contestants won multiple honors in the fierce competi-Quality technology training
external training sessions were organized during the reporting tion inspiring all quality inspection personnel in the
period covering 1838 participants. This has continuously enhanced Company to refine their skills and jointly build a line of
the understanding and execution level of inspection personnel defense for drug quality and safety.regarding the new standards ensuring that inspection work Provincial Drug Quality Inspector
Professional Skills Competition
complies with the latest regulatory requirements.
47 48About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability Product System for Value
Group Sustainability Management
Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability ReportCreation
Chairman Stable Growth Management Shared Future
Impact Risks and Opportunities Indicators and Objectives
In response to the high requirements of the pharmaceutical sector for product safety compliance and risk controllability the Focusing on its established quality objectives the Company continuously monitors and evaluates key quality indicators. By
Company has integrated product quality-related impacts risks and opportunities into a unified quality management framework. improving risk prevention and control measures and operational management mechanisms it promotes the continuous improve-
This framework covers key stages such as R&D production supply chain distribution and post-market management forming a ment and effective operation of its quality management system ensuring the implementation of quality objectives and phased
risk management process that spans the entire product lifecycle. By embedding quality risk management requirements into its management requirements.daily quality operations the Company continuously identifies factors that may affect product quality drug safety operational
stability and market trust thereby promoting a continuous shift in quality management towards proactive identification and
in-process control. During the reporting period the Company
The Company systematically identifies quality-related risks and potential impacts through channels such as the execution of No major quality accidents throughout the year non-conformance announcements
quality management systems production process control supplier quality management pharmacovigilance information monitor-
ing and internal and external audits. It conducts graded assessments based on the likelihood and severity of the risks. For major ? time ? item
or urgent quality risk events the Drug Safety Committee coordinates the assessment the Quality Management Department takes
pass rate in supervisory inspections product release pass rate
the lead and relevant business departments participate collaboratively to ensure that risk information is identified effectively
judged and responded to in a timely manner. During the reporting period the quality audit coverage rate for pharmaceutical ???% ???%
manufacturing enterprises R&D enterprises and non-pharmaceutical business-related enterprises was 100%.Pharmacovigilance
Internal quality monitoring Customer complaints Regulatory spot Adverse reaction Drug safety
checks monitoring information The Company conscientiously fulfills its primary responsibility as a Marketing Authorization Holder treating pharmacovigilance as
a crucial lever for ensuring public medication safety. It strictly adheres to regulations such as the Measures for the Reporting and
Monitoring of Adverse Drug Reactions and Good Pharmacovigilance Practice. It has established a pharmacovigilance center
staffed with full-time professionals to uniformly coordinate and supervise pharmacovigilance activities. A pharmacovigilance
management platform has been developed to unify systems standards operational procedures and assessment requirements. It
Production process Product adverse also conducts regular supervision inspection and guidance for its subsidiary enterprises and drug research institutions to ensure
supervision product After-sales calls Spot checks by Adverse drug reaction r e a c t i o n / d r u g that all pharmacovigilance activities are conducted in compliance with regulations.and material inspec- official website drug regulatory monitoring system safety information
tion deviation identi- store feedback authorities customer complaints obtained by compa- The Company continuously collects drug safety information through multiple channels including the National Adverse Drug
fication ny employees Reaction Monitoring System customer complaints feedback from medical institutions and literature monitoring. Upon receiving
information about adverse reactions it promptly records assesses causality and implements follow-up management. All informa-
tion meeting reporting criteria is classified and submitted within specified timeframes via the national direct reporting system for
adverse drug reactions in accordance with regulatory requirements. Based on regular analysis and assessment results it dynami-
cally optimizes risk prevention and control measures to enhance the timeliness and effectiveness of its risk response.Channels for identifying quality issues
At the same time the Company continues to strengthen its professional capabilities in pharmacovigilance. Focusing on new
regulations and key inspection points it organizes specialized pharmacovigilance training and incorporates course resources
In terms of risk response and control the Company implements closed-loop management of quality abnormalities and potential from the National Center for ADR Monitoring China and professional industry institutions achieving full coverage of personnel in
risks through mechanisms such as deviation management change management and corrective and preventive actions (CAPA). relevant positions. This continuously enhances professional judgment and compliance execution capabilities providing strong
The Company analyzes the root causes and impact levels of risks and takes corresponding disposal measures based on the risk support for the ongoing management of drug safety risks.level to prevent the escalation or recurrence of risks. By tracking the effectiveness of corrective actions it continuously optimizes
its quality management systems and processes steadily enhancing its overall quality management capabilities.While strengthening quality risk prevention and control the Company also focuses on the positive impacts and development
opportunities brought by enhancing its quality management capabilities. By continuously improving its quality management
system and enhancing product quality consistency and full-process compliance the Company strengthens its adaptability to
changes in regulatory policies solidifies its foundation of market trust and reduces the potential impact of quality issues on its
operations and finances from the source.
49 50About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Customer Rights and Interests and Service Assurance Case Professional service capability building for DTP pharmacies
In 2025 Renmintongtai continued to refine its DTP training system
in alignment with the professional service requirements of DTP
To ensure public medication safety and enhance service accessibility the Company has established and implemented internal pharmacies. It organized targeted training sessions led by physi-
policies such as the Customer Service Management System. It embeds customer service requirements into product information cians from Grade A tertiary hospitals and senior licensed pharma-
management service response and daily operations to promote standardized and orderly customer service activities effectively cists employing a blended approach combining online and offline
safeguarding the legitimate rights and interests of customers and consumers. Aligned with its corporate culture and values it learning with self-study. The training covered multiple key disease
unifies customer service standards and codes of conduct clarifying a service orientation centered on integrity professionalism areas and core business modules strengthening pharmacists'
collaboration and efficiency. It continuously cultivates a professional standardized pragmatic and efficient service image to professional capabilities in full-cycle patient management
enhance brand reputation and customer satisfaction. cold-chain drug handling standards and compliant service delivery.Concurrently the Company regards professional capability development as a critical enabler. Focusing on key service scenarios Building on this foundation Renmintongtai innovatively introduced
such as rational drug use guidance chronic disease management support and special medication assurance it continuously an AI-powered intelligent learning and practice platform with drug
enhances the professional competence and service capabilities of front-line staff driving the evolution of customer service from consultation and patient follow-up as core training scenarios.basic support toward more professional refined and sustained assistance. Through routine knowledge exercises and simulated follow-up
drills the platform helps pharmacists consolidate professional
knowledge on drug indications dosage and administration
standardize service procedures and communication approaches
Case Special topic training on chronic disease management to enhance frontline pharmacy service capabilities and enhance the professionalism and consistency of patient
follow-ups.Renmintongtai periodically organizes training on key disease management focusing on the long-term medication
and health management needs of patients with chronic diseases. These sessions concentrate on high-prevalence
chronic conditions such as diabetes systematically explaining disease characteristics key points of standardized AI-Powered Intelligent Learning
medication use and related health management content to enhance the professional service capabilities of and Practice Platform
front-line employees.In October 2025 Renmintongtai invited a chronic disease management expert to deliver a special training session
on Chronic Disease Management for Diabetes covering disease mechanisms complication risks individualized
medication and lifestyle interventions. Sixty front-line store employees participated effectively enhancing their
practical competencies in chronic disease identification medication guidance and patient communication.The Company has established the Quality Complaint Manage-
ment System. When customer complaints or quality-related During the reporting period the Company
issues arise it follows established procedures for acceptance
investigation resolution feedback and corrective action. A
tiered management approach is applied based on the nature and
Handled ???% customer feedbacks
severity of the issue with clear assignment of responsible parties
and handling requirements. Relevant personnel promptly identify
root causes implement corrective measures and track their
effectiveness promoting closed-loop issue management and
continuous improvement of service quality to prevent recurrence
of similar issues.Considering store layouts and actual service scenarios the Company is gradually exploring the appropriate extension of conve-
nient services without compromising the standardized operation of its core business. Selected stores provide essential conve-
nience support and basic assistance to residents of surrounding communities and outdoor workers enhancing service accessibil-
ity and enabling stores to function not only as healthcare access points but also as community service hubs. By organically
integrating customer service with quality management terminal operations and community engagement the Company continu-
ously shifts its customer relationships from transactional interactions toward more stable and sustainable service partnerships
Training on chronic disease management for diabetes systematically addressing the diverse needs of different groups.
51 52About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Hapharm Performance in Special Topic to Solidify a Foundation for Product System for Value
Employee Development
Report from the Management Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth
Creation Management Shared Future
In advertising and communications management the Company implements a unified content review system and a tiered account-
Case Consumer training and interactive service programs ability framework. All advertising and promotional materials must undergo internal review and filing. Promotions involving pharma-
ceuticals or health-related content are subject to statutory review requirements and pre-publication compliance and risk assess-
ments. In media collaborations and new media communications it adheres to standardized expression and cautious publishing
The Company continuously implements consumer training and interactive service programs. Through standardized
practices to prevent inappropriate promotion and compliance risks. At the point of service the Company relies on its retail stores
health education and service activities it enhances public health literacy and terminal service quality extending
to uniformly implement standardized service processes and pharmaceutical care requirements guiding consumers toward rational
customer engagement beyond transactional interactions toward long-term service relationships.and safe medication use through structured medication counseling and service management thereby avoiding misleading recom-
mendations or improper marketing practices.The program primarily leverages chain pharmacies as its main platform complemented by community and public
cultural venues to establish a multi-scenario service model. Through patient education health lectures and interac-
Additionally the Company emphasizes the social value of health communication disseminating content on drug safety rational
tive experiences it delivers scientific and standardized knowledge on medication use and health management to
drug use and health science through official channels to enhance public health awareness. To meet advertising compliance and
consumers. The Company collaborates with chain partners on activity planning and execution to enhance store
service standardization requirements it regularly organizes training on relevant regulations and practical implementation cover-
service capabilities and accessibility. In 2025 the program conducted a total of 405 sessions reaching approximate-
ing key areas such as advertising production and review procedures official website and new media content operations and
ly 13000 consumers. While improving service engagement and consumer satisfaction it also accumulated practical
promotional standards for live-streaming and e-commerce platforms. This further strengthens compliance awareness and risk
experience for refining the terminal service system and advancing health science communication.mitigation capabilities among relevant personnel promoting more standardized and orderly external communications and terminal
services.Case Advertising compliance review and regulatory training
In May 2025 to further enhance the standardization and security of its external communications the Company
organized "Training on Advertising Compliance Review and Relevant Laws and Regulations" delivering systematic
instruction to key personnel involved in external promotion through a blended online and offline format. The training
addressed critical topics including advertising production and review procedures official website content operation
standards publishing requirements for official WeChat accounts and self-media platforms live-streaming content
management and promotional language standards for e-commerce platforms. It also provided focused guidance on
Consumer training and interaction advertising review essentials and common risks contextualized within real-world work scenarios.The training covered 105 personnel from
relevant departments effectively enhancing
Case Emergency cold chain distribution to ensure the medication needs of medical institutions their understanding and execution of adver-
tising compliance requirements strengthen-
ing risk identification and compliance aware-
In April 2025 the Yichun Maternal and Child Health Hospital encountered an urgent medication need during clinical ness in external communications and provid-
treatment that could not be met by its in-house inventory in the short term. Upon receiving the request Logistics ing assurance for the standardized and order-
Center immediately activated its emergency customer service and distribution response mechanism coordinating ly conduct of the Company's marketing and
logistics warehousing and quality functions to swiftly complete drug allocation verification cold-chain packaging communication activities.and dispatch. During transportation the Company strictly adhered to cold-chain transport and temperature control
requirements arranged dedicated direct delivery and implemented end-to-end monitoring to ensure the drugs met
usage standards for both delivery timeliness and quality safety. The required medications were delivered on time and
safely ensuring continuity of clinical treatment. Responsible marketing training
Responsible Marketing
During the reporting period the Company
The Company places consumer health and legitimate rights at the core of its operations and upholds responsible marketing as a
fundamental principle of its market activities. It continuously standardizes its marketing and external communication practices. In
all marketing communication activities it strictly complies with laws and regulations including the Advertising Law of the People's
Republic of China the Drug Administration Law of the People's Republic of China and the Law of the People's Republic of China Performed ?? responsible marketing audits
on the Protection of Consumer Rights and Interests. It has established a marketing compliance management mechanism covering
the entire process—from planning and review to publication—to ensure all content is truthful accurate and objective without
exaggerating product efficacy inducing health anxiety or misleading consumers.
53 54About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth
Creation Management Shared Future
The Company continues to advance its transparent procurement system leveraging a unified SRM (supplier relationship manage-
Supply Chain Management ment) platform to enable end-to-end digital management of suppliers and ensure full traceability. Procurement activities strictly
and Security adhere to the principles of openness fairness impartiality full competition and compliance & integrity ensuring equal access to procurement information for all qualified suppliers. The procurement process is fully traceable and auditable effectively mitigating
risks of improper transactions and enhancing transparency and operational efficiency in procurement management.Case Digital platform empowering transparent procurement and industrial chain collaboration
In accordance with GMP and other applicable laws regulations and industry regulatory requirements the Company has estab-
lished and continuously improved its supplier management system. Through policies including the Supplier Management Regula- To enhance standardization and transparency in procurement and sales and improve industrial chain collaboration
tions Detailed Rules for Supplier Sourcing Management and Management Procedures for Drug Supplier Evaluation Standards it efficiency the Company has advanced its supply chain management through digital transformation establishing an
standardizes key processes—sourcing onboarding collaboration evaluation and exit—providing institutional assurance for the integrated upstream-downstream management system centered on its enterprise-level SRM (Supplier Relationship
security compliance and stable operation of the supply chain. The Board of Directors oversees supply chain strategy; senior Management) system and the SAP Hybris distributor platform. The dual-platform architecture enables seamless data
management coordinates strategic implementation; and the Tendering and Procurement Department is responsible for execution integration across the entire procurement and sales chain offering a unified and open collaborative environment for
including supplier onboarding evaluation and risk control. upstream and downstream small and medium-sized enterprises (SMEs). As of the end of the reporting period the
platforms served over 20000 upstream suppliers and more than 4000 downstream commercial customers with SMEs
accounting for over 90%. Platform adoption has significantly improved operational efficiency: order processing
Information manage- Evaluation and efficiency increased by over 46% procurement cycles shortened by approximately 30% contract signing efficiency Sourcing Onboarding
ment classification rose by 80% and inventory data accuracy reached 99.8%. Through centralized procurement and compliance verifica-
Search for potential Potential suppliers register Maintain unified Conduct regular (at least tion mechanisms the Company has reduced procurement costs minimized inventory accumulation and continuously
suppliers through various and submit qualification supplier master data annually) or ad hoc evalua- enhanced the standardization and security of supply chain operations.channels (e.g. internet documents on the SRM and database informa- tions of qualified suppliers
exhibitions industry platform. After passing tion (potential across multiple dimensions—
associations). review—which may include qualified blacklist) on including quality delivery The Company considers integrity and compliance as a fundamental prerequisite for supplier cooperation strictly adhering to
product testing and on-site the SRM platform. price service and overall national laws and regulations and company policies. It maintains a "zero-tolerance" policy towards commercial bribery and
inspections—they complete capability—and apply improper trading practices. During the bidding and cooperation processes the Company uniformly requires suppliers to sign the
the onboarding process per dynamic classification HARBIN PHARMACEUTICAL GROUP CO. LTD. Bidder Integrity Commitment Letter. Performance under this commitment is
the introduction plan and (strategic leverage routine integrated into the supplier evaluation and management system serving as a crucial basis for continued cooperation. Through
become qualified suppliers. bottleneck) and grading (A institutional constraints and process controls integrity requirements are embedded into key aspects such as supplier onboarding
B C D) based on results. bidding and procurement and contract performance management preventing integrity risks in the supply chain from the source
and ensuring the fairness and compliance of procurement activities.Relationship manage- Performance manage-
Exit and reinstatement
ment ment
Suppliers failing evaluation Establish communication Engage with support and During the reporting period the Company
criteria or encountering mechanisms handle mutual address suppliers experienc-
major issues are removed complaints and provide ing issues in quality delivery
from the qualified supplier support to suppliers with or service (including issuing
list or placed on the blacklist. low ratings. warnings suspending
Reinstatement after exit cooperation downgrading or Achieved ???% signing rate for the Integrity Commitment Letter among bidding suppliers
requires re-completion of the blacklisting).onboarding process.Supplier management process
Supply Chain Risk Management
During the reporting period the Company
The Company embeds risk management into the full-lifecycle management of its suppliers. By controlling access monitoring
Total number of suppliers Proportion of local suppliers Local procurement ratio processes and making dynamic adjustments it systematically identifies and addresses compliance and operational risks within
the supply chain. Prior to cooperation it conducts audits focusing on key requirements such as compliant operations quality and
safety and business ethics to prevent high-risk entities from entering its supply system. During cooperation it continuously
???? ??.??% ??.??% tracks supplier operational performance by integrating fulfillment records issue feedback and audit results and takes timely
management actions in response to evolving risks to prevent their accumulation and spread.
55 56About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability ReportCreation
Chairman Group Sustainability Stable Growth Management Shared Future
Relying on internal audits and special inspections the Company supervises and verifies processes and key risk points related to
Supply chain risk management goals
supplier management. Risks identified through audits are incorporated into subsequent management and improvement cycles
strengthening closed-loop risk prevention and control. By integrating information security and integrity requirements into audit
and process management it ensures the standardization transparency and controllability of supply chain operations. Annual audit coverage for tier 1 suppliers ≥ 95% ESG special audit coverage ≥ 50%
A supplier risk warning response time of ≤ 24 hours
During the reporting period the Company
Conducted document audits for Conducted on-site audits for Had ?? suppliers that underwent and passed
???? suppliers ??? suppliers environmental and social audits Sustainable Supply Chain Management
The Company continues to advance the development of a sustainable supply chain by leveraging policy documents such as the
Supplier Management Regulations and the supplier full-lifecycle management mechanism. It incorporates sustainability require-
ments—including environmental protection product quality work safety labor practices and business ethics—into supplier
onboarding cooperation and evaluation processes. Through policy documents and procurement contract clauses it clearly
defines supplier responsibilities and obligations regarding compliant operations anti-commercial bribery and information protec-
Case tion. Suppliers found to have committed serious legal violations posed quality and safety hazards or demonstrated commercial Construction of GAP bases for core Chinese materia medica at Shiyitang Chinese Medicine dishonesty are subject to restrictions or termination of cooperation in accordance with applicable laws and regulations. During
cooperation the Company integrates factors such as quality and safety performance fulfillment reliability and compliance
To strengthen source-level quality management of the traditional Chinese medicinal materials supply chain and records into supplier evaluation and tiered management using evaluation outcomes to drive corrective actions and continuous
ensure the stability safety and traceability of core Chinese materia medica raw materials Shiyitang Chinese improvement thereby promoting responsible supplier conduct and enhancing management capabilities to steer the supply chain
Medicine focuses on building good agricultural practice (GAP) bases for Chinese materia medica continuously toward greater standardization and sustainability.advancing standardized cultivation and quality control systems for authentic medicinal materials.The Company conducts supplier training centered on sustainable During the reporting period the Company
Shiyitang Chinese Medicine focuses on core varieties including Acanthopanax senticosus Lonicera japonica supply chain management requirements to reinforce understanding
Forsythia suspensa Scutellaria baicalensis and Schisandra chinensis collaborating with multiple Chinese materia and implementation of quality compliance and responsibility
medica cultivation enterprises to jointly build and share GAP bases thereby advancing quality management require- standards fostering supply chain collaboration and enhancing Performed ???supplier
ments upstream to the cultivation stage. Relevant bases have successively passed the GAP compliance extension sustainability capabilities. training sessions
inspections conducted by the Heilongjiang Provincial Medical Products Administration. Among them the cumulative
inspected area for cultivation bases of Lonicera japonica Acanthopanax senticosus Forsythia suspensa and Scute-
llaria baicalensis exceeds 18000 mu achieving controllable sourcing manageable processes and traceable quality
for major Chinese materia medica raw materials.Equal Treatment of Small and Medium-Sized Enterprises
The Company upholds equal treatment of small and medium-sized enterprises (SMEs) in supplier management and procurement
practices. Its bidding and procurement management system explicitly prohibits discriminatory or exclusionary conditions that
restrict SME participation ensuring they enjoy fair competition opportunities in accordance with the law.The Company publicly discloses procurement information via its official website and third-party platforms ensuring all suppliers—
including SMEs—have equal access to procurement information understand procedures and can participate in competitive
bidding. Additionally the Company has established a standardized settlement and payment mechanism and strictly fulfills
contractual payment terms.and sustainable supply chain cooperation ecosystem.GAP cultivation bases
57 58About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon Appendix 2025 Sustainability ReportGroup Sustainability Stable Growth Creation
to Build a Safe and
Chairman Management Shared Future
Information Security During the reporting period the Company
and Privacy Protection
Conducted attack-defense and emergency response drills
?
Had confirmed data security incidents
The Company continuously enhances its information security and privacy protection governance system by establishing a
dedicated network and information security management organizational structure including a Network and Information Security ?
Leading Group and an Emergency Response Working Group. These bodies coordinate decision-making on major information
security and privacy matters and provide unified command and coordination for emergency responses to cybersecurity incidents Had confirmed customer privacy breach incidents
ensuring orderly and efficient risk response. ?
At the policy level the Company continuously refines its information security and privacy protection framework centered on the Information security skills training
Regulations on the Management of Computer Use and Information Systems which clearly define norms for information system
usage allocation of information security responsibilities and requirements for data access and use—providing institutional assur-
ance for stable system operations and compliant data management. Concurrently the Company strictly implements China's
Cybersecurity Classified Protection requirements organizing filing and assessment activities for its information systems. Its
official website collaborative office system and integrated business-finance system have obtained Level II and Level III Classified
Protection certifications and the Company continuously elevates its overall security posture through third-party evaluations. Medicine Accessibility Enhancement
In technical protection and operational management the Company strengthens cybersecurity defenses and emergency response
capabilities based on the actual operating conditions of its information systems. Through implementing cybersecurity initiatives
conducting attack-defense drills and performing phishing email simulations it continuously assesses system resilience and Governance
employee security awareness promptly identifying and remediating potential vulnerabilities to enhance overall defensive and
incident response capabilities. Medicine accessibility is a key topic in safeguarding public health and well-being and constitutes a core component and critical
practice area of the Company's social responsibility. Its governance level and implementation effectiveness are directly linked to
The Company enforces strict privacy management for customer and patient information. Such information is collected only in the fairness of medical resource allocation the availability and affordability of pharmaceutical services and ultimately the realiza-
legitimate and compliant business contexts and centrally managed through a unified system platform with clear requirements for tion of societal health outcomes. To this end the Company leverages a collaborative mechanism among multi-level governance
data storage access control and confidentiality. Retention dissemination or disclosure of this information in non-work-related bodies to continuously strengthen the governance foundation in medicine accessibility systematically translating governance
contexts is prohibited. Upon personnel role changes or departures the Company strictly enforces information handover and data requirements into actionable measures. Through systematic governance it empowers core objectives such as the decentralization
clearance protocols to ensure information security remains under control. of medical resources and enhanced accessibility of innovative medicines thereby demonstrating its commitment to public health
stewardship.During the reporting period Renmintongtai further revised the Management System and Standard Operating Procedures for
Specialty Pharmacies adding specific patient privacy protection requirements. The revision mandates adherence to the principle
of data minimization in information collection and requires that patients be fully informed—at the time of initial service—about the Strategy
purpose scope retention period and protective measures related to data collection with informed consent obtained accordingly.It also refines standards for data desensitization and strengthens management requirements for information storage use and Risk name Specific description Time horizon1 Financial impact Response measures
transmission thereby further safeguarding patient rights and enhancing service compliance and public trust.Simultaneously the Company continues to strengthen awareness and training on information security and privacy protection by
integrating management requirements into daily workflows through thematic training sessions internal platform communications The drug pricing mechanism is
and cautionary education. During the reporting period it published 42 issues of cybersecurity awareness content and organized Strengthen price influenced by multiple factors—
security awareness and skills training programs focused on information system operations and business scenarios continuously management and market including national centralized Increased revenue
Price
reinforcing the foundational management of information security and privacy protection. Short and monitoring and formulate procurement policies market volatility and greater
instability risk medium term pricing strategies that
competition dynamics and profit uncertainty
align with cost changes
fluctuations in raw material costs—
and market demand
resulting in certain price volatility
1Impact duration refers to the time scope of material impacts on the Company. Short-term: within 1 year; medium-term: 1 to 5 years;
long-term: more than 5 years.
59 60About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon Appendix 2025 Sustainability ReportGroup Sustainability Stable Growth Creation
to Build a Safe and
Chairman Management Shared Future
Risk name Specific description Time horizon Financial impact Response measures
Case Focusing on full-course disease management to aid patient recovery
The pharmaceutical supply chain In November 2025 the Company hosted a lung cancer
encompasses multiple segments—
Optimize the supply chain patient education event titled "Foreseeing the Future
production warehousing logistics
Decreased sales and channel network and Creating a New Life Together" at its DTP Management
Poor channel and last-mile distribution—with Short and
revenue and higher enhance supply assurance Center attended by 22 patients. A chief physician from a
distribution risk uncertainties in channel coordina- medium term
operating costs for key regions and critical tumor hospital was invited as the keynote speaker to
tion and delivery efficiency; supply
products
assurance capacity in certain share professional insights on key aspects of full-course
regions requires improvement disease management helping patients and their families
develop a scientifically informed understanding of the
condition. The event included one-on-one consultation
sessions to facilitate discussions between patients and
healthcare providers about treatment concerns. In collab-
oration with physicians the pharmacy established a
Opportunity type Specific description Time horizon Financial impact Response measures fast-track consultation channel to streamline follow-up
procedures and address access barriers. This event
provided professional guidance and services created a
Exclusive patient education event
platform for doctor-patient communication and support- for lung cancer patients
Evolving public health needs and ed standardized treatment and long-term health manage-
growing demand for affordable
Advance product portfolios ment for lung cancer patients.highly adaptable medicines in Increased revenue
Pharmaceutical tailored for grassroots and
grassroots and emerging markets Long-term and higher cash
innovation emerging markets and
continue to expand product inflows
enhance market coverage
application scenarios and market In 2025 the Company further deepened its "Internet + Pharmaceutical Health" service model with a core focus on enhancing
opportunities medication purchasing convenience and upgrading health service experiences. It comprehensively optimized and scaled its O2O
business system across chain stores. Leveraging the physical advantages of its offline store network and the technological
enablement of its digital platforms the Company successfully established an integrated "online + offline" service loop efficiently
meeting consumers' health and medication needs across all times and scenarios significantly expanding service reach and
response efficiency.Access Channels
Case Empowering science communication upgrades through expert livestreaming to improve public health literacy
The Company has deep expertise in the pharmaceutical distribution sector and has built an efficient province-wide delivery
network leveraging high-quality channel resources supported by strong market coverage and marketing capabilities. It has estab- In July 2025 the Marketing Company built a nutrition
lished a comprehensive terminal customer coverage system encompassing graded hospitals primary healthcare institutions and popular science platform via short videos. Aiming at
retail pharmacies. Long-standing cooperative relationships with key medical institutions and commercial clients are reinforced public dietary misunderstandings and nutritional
through continuous service quality improvements. In its retail segment the Company focuses on refined operations and optimized needs it converted professional nutritional knowledge
layout of directly operated stores while utilizing DTP specialty pharmacies1 and hospital-adjacent stores as key channels to into accessible and widely-spreadable content to lower
capture prescription outflow delivering professional and standardized pharmaceutical services to consumers. the public’s threshold for acquiring health knowledge.To guarantee content quality a special team was estab-
As of the end of 2025 the Company operated 357 directly managed retail stores including 226 stores in Harbin and 131 stores lished for content planning and production based on
outside the city. The Company had a total of 15 DTP specialty pharmacies to better meet patients’ medication needs. The DTP basic nutrition and real-life scenarios. Up to now over
pharmacies cumulatively served 250000 patients with follow-up services covering nearly 190000 visits. 40 nutrition popular science short videos have been
released attracting more than 60000 user interac-
tions which has effectively expanded popular science
coverage and improved public health literacy.
1DTP pharmacies (Direct to Patient) refer to specialized pharmacies through which pharmaceutical manufacturers or distributors provide prescription
drugs—primarily specialty and innovative medicines—directly to patients. Employee Live Broadcast Column
61 62About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability Special Topic to Solidify a Foundation for Product System for Value Management Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability ReportCreation
Chairman Group Sustainability Stable Growth Management Shared Future
Inclusive Healthcare Impact Risk and Opportunity Management
Product pricing basis
The Company systematically identifies and manages impacts risks and opportunities related to medicine accessibility in pursuit
of its goal to ensure public access to essential medicines. Considering evolving policy landscapes market feedback and opera-
The Company adopts a market-oriented pricing mechanism conducting nationwide competitive price surveys to tional performance it continuously monitors factors—including drug price volatility supply channel efficiency and coverage in
systematically understand market price baselines and regional patient affordability thresholds. Combined with grassroots and remote areas—that may affect medicine availability and affordability. Comprehensive assessments are conducted
full-chain cost accounting of product manufacturing it aligns pricing with market realities and ensures patient across dimensions such as patient affordability supply reliability and service reach. For key identified risks the Company imple-
affordability fulfilling its corporate mission. ments ongoing management through pricing justification channel network optimization and service model enhancements
supported by dynamic tracking via operational data and market insights. While mitigating accessibility risks it seizes opportuni-
ties arising from innovative drug adoption channel expansion into lower-tier markets and service model innovation driving more
equitable and effective delivery of pharmaceutical resources to meet societal needs.Pricing data and changes for key products
Pricing of the key product Nao An Pian from Hayao No.6 Factory follows centralized procurement policy guidance.In 2023 it was successfully awarded a contract in the National Traditional Chinese Medicine Procurement Alliance's Indicators and Objectives
volume-based procurement program with winning bid prices of RMB 22.41 per box (24-tablet pack) and RMB 33.12
per box (36-tablet pack)—a 17% reduction that effectively alleviates patient medication costs. This price has been Target
progressively implemented across provinces since September 2023. Following the completion of a new bidding
round in 2025 the same price remains in effect nationwide strongly supporting inclusive pricing objectives.Launched products are timely registered on provincial procurement platforms nationwide to achieve
Fair pricing strategy
rapid terminal sales coverage at all levels of medical and health institutions across the country.A comprehensive pricing justification mechanism has been established covering analysis of market competition
dynamics competitive price benchmarking invoicing price calculation and compliance validation. Through multi-di-
mensional systematic review pricing strictly adheres to national pharmaceutical pricing policies while maximizing
patient benefits effectively reducing financial burdens and reflecting a commitment to inclusive health access.The Company actively participates in national volume-based procurement and relevant policies includ-
ing the "Three Entries" initiative. It effectively improves public access to medicines and reduces
Case Responding to the hierarchical diagnosis and treatment policy to help enhance medication accessibility residents’ economic burden of medical treatment and medication.Promoting hierarchical medical care and improving access to medicines at grass-
roots level has been a top priority of China’s medical and health work in recent
years. The state has continuously introduced relevant policies including incorpo-
rating village clinics into medical insurance pooling advancing the "Three Entries"
of centralized procurement improving compact county medical communities and
realizing grassroots medication linkage.The Company deeply participates in national volume-based procurement and the
National Chinese Patent Medicine Alliance Procurement. Its products have been
selected in multiple batches of such procurement projects. The substantially
reduced selected prices effectively meet the medication demands of medical
institutions and ease patients’ medication burden.Regarding the "Three Entries" policy the Company actively applies for relevant
product listings. Nao’an Tablets Fosfomycin Sodium for Injection Ceftriaxone
Sodium for Injection Cefazolin Sodium for Injection and Cefradine Capsules have
been included in the provincial "Three Entries1" catalogues of multiple regions.
1Refers to the access of centralized-procurement medicines to grassroots medical institutions private medical institutions and retail pharmacies.
636403
Green Operations to
Advance Low-Carbon
Management
The content of this chapter
Environmental Compliance Management ??
Climate Change Tackling ??
Energy and Resource Management ??
Pollutant Emissions ??
Waste Management ??
Ecosystem and Biodiversity Conservation ??About This Message About 2025 Key Governance and Compliance Green Operations to
from the Hapharm Performance in
Sustainability Special Topic to Solidify a Foundation for Product System for Value
Employee Development
Report Management Advance Low-Carbon Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation
to Build a Safe and
Management Shared Future
Environmental Compliance Management Case The Company’s organization of specialized environmental protection training to strengthen the green compliance line of defenses
In September 2025 the Company led a specialized
The Company strictly complies with relevant laws and regulations including the Environmental Protection Law of the People's environmental protection training session involving
Republic of China the Law of the People's Republic of China on the Prevention and Control of Environmental Pollution by Solid relevant personnel from its subsidiaries with a total of 30
Waste the Air Pollution Prevention and Control Law of the People's Republic of China and the Water Pollution Prevention and participants. This training was closely aligned with the
Control Law of the People's Republic of China. It has formulated and implemented the Hapharm Group Environmental Protection current severe environmental regulatory situation and
Management Measures. The Company continuously advances pollution prevention and control efforts to ensure stable and precisely met the Company's actual needs for environmen-
compliant emissions of all pollutants. With a core focus on fulfilling its primary environmental responsibilities and strengthening tal management. The content covered core modules such
end-to-end supervision it steadily progresses in building a green manufacturing system. Its subsidiary Sanjing Mingshui was as in-depth interpretation of environmental laws and
awarded the National Green Factory designation in 2024. regulations analysis of typical environmental violation
cases practical methods for corporate environmental
The Company has established a tiered environmental management responsibility system designating the principal executives of self-inspection and emergency response plans for
each subordinate production enterprise as the primary persons accountable for environmental protection. It centrally coordinates environmental incidents. The training effectively strength- The Company's environmental protection training
annual internal audits of the environmental management system and maintains end-to-end oversight to ensure responsible ened the environmental compliance awareness and sense
entities complete closed-loop corrective actions for any weaknesses or risks identified guaranteeing the system's sustained and of responsibility of the participants from all subsidiaries
high-quality operation. systematically enhancing their capabilities in environmen-
tal risk identification hazard investigation and emergency
The Company has formulated and issued the Emergency Response Plan for Environmental Emergencies aiming to effectively response laying a solid foundation for the Company to
respond to environmental emergencies triggered by various production safety accidents and natural disasters. This plan is continuously strengthen its environmental management
designed to prevent the diffusion of pollutants and toxic and hazardous substances into the atmosphere water bodies soil and line of defense and achieve green and compliant opera-
other environmental media outside the plant boundaries thereby minimizing the negative impact on the environment. At the same tions.time the Company organizes environmental protection training conducts emergency drills and establishes an effectiveness
evaluation mechanism to ensure the operability and effectiveness of the plan. Additionally the Company continuously improves
its long-term environmental management mechanisms regularly carries out environmental hazard investigation and rectification
work ensures effective investment in hazard rectification funds and prevents the occurrence of environmental risk incidents. Climate Change Tackling
During the reporting period the Company Governance
Conducted??emergency drills for environmental risk incidents The Company deeply implements the national "dual carbon" strategic deployment incorporating climate change tackling into its
core corporate governance and steadily promoting energy conservation and carbon reduction efforts based on its existing
Organized????participants for emergency drills of environmental risk incidents management structure. In terms of carbon emission governance the Company has established standardized working mechanisms
for Scope 1 and Scope 2 carbon emission inventories. It conducts comprehensive inventories and precise accounting according
Conducted ??environmental management training sessions to a set cycle while simultaneously sorting archiving and verifying carbon emission data to ensure its authenticity and traceabili-
ty. Simultaneously it dynamically optimizes energy conservation and carbon reduction management measures based on the
Conducted environmental management training with a total duration of ???? hours inventory results promoting the effective integration of governance requirements with all aspects of production and operation
and continuously enhancing its governance effectiveness in response to climate change.Conducted environmental management training with an average duration of ?.?? hours per person
Organized????participants for environmental management training
Organized???participants for environmental protection publicity and public welfare activities
Invested RMB??.????million in environmental protection
?Scope ? refers to direct GHG emissions from sources owned or controlled by the Company mainly including boiler combustion vehicle exhaust etc.;
Scope ? refers to indirect GHG emissions from the generation of purchased and consumed external energy such as electricity steam heat and
cooling.
67 68About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability
Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Strategy
Risk type Specific description Time horizon Financial impact Response measures Specific description Specific description Time horizon Financial impact Response measures
Integrate extreme weather
risks into operational and Align with evolving
Production
emergency management market demands and
Extreme weather events may Emerging disease prevention stoppage
systems enhance production business strategy
disrupt production sched- needs and public health Extreme Short and losses and
continuity safeguards and Revenue growth continuously monitor
weather ules and logistics arrange-
Emerging market challenges driven by climate
medium term increased Long-term
contingency plans and and expanded climate-related health
ments opportunities change may stimulate demand operating costs
strengthen risk identification market share trends and drive a for related therapies and
and response capabilities for rational portfolio layout products
Phys- critical nodes
of products and
solutions
ical
risks
Enhance energy consumption
Rising ambient temperatures
Higher energy analysis and cold chain
Increase in impose stricter requirements
costs and operations management
annual on temperature control and
Long-term increased promote energy-saving The Company focuses on strengthening its management foundation through capacity building. In response to climate change-re-
average cold chain management across
operating technologies and equipment
temperature production storage and lated risks and emission reduction requirements and in line with its production and operational realities the Company organizes
expenses optimization and improve
transportation relevant training to deepen the understanding of climate change topics and related compliance requirements among management
energy efficiency personnel and employees in key positions thereby enhancing their ability to identify and respond to climate-related risks in daily
management.Continuously monitor
evolving climate and
Carbon emissions and energy
environmental policies
management regulations are Increased
integrate relevant require- Case Participating in special topic training on energy conservation and carbon reduction to progressively tightening compliance
Medium and enhance low-carbon management capabilitiesments into environmental
Policy risk subjecting the company to costs and
long term management and business
heightened compliance higher capital
decision-making processes In 2025 the Company organized relevant
obligations and management expenditures
and proactively plan energy personnel to participate in a special topic
pressure
conservation and carbon training on energy conservation and carbon
reduction initiatives reduction held by the relevant departments of
Songbei District Harbin City Heilongjiang
Conduct technical Province. The training centered on the require-
Higher assessments and phased
Existing energy equipment ments of the "dual carbon" goals systematically
investment in optimization of energy
and production facilities explained basic energy theories energy
Tran- equipment equipment and production
Technology face pressure to upgrade to Medium and consumption indicator accounting methods and
sition upgrades and facilities implement
risks meet current energy long term key points of carbon peak and carbon neutrality
Risk increased energy-saving retrofits
efficiency and emission
depreciation aligned with operational policies helping participants to enhance their
standards
expenses realities and enhance asset understanding of the policy requirements and Special topic training on energy conservation
utilization efficiency implementation paths for energy conservation and carbon reduction
and carbon reduction.Strengthen environmental
Environmental governance
management and disclo-
and climate response
sure continuously advance
performance attract Higher
energy conservation
Reputational heightened attention from the Short and financing costs
carbon reduction and At the same time the Company focuses on optimizing its energy structure in the production and operation process. It explores
risk public and investors medium term and reduced
environmental compliance the application of clean energy sources such as photovoltaic power generation based on its energy consumption conditions. By
increasing corporate revenue
practices and enhance increasing the proportion of renewable energy source usage it gradually reduces the emission pressure brought by traditional
reputation management
external transparency and energy consumption.pressure
management credibility
69 70About This Message About 2025 Key Hapharm Performance in Sustainability
Governance and Compliance Green Operations to Employee Development
Report from the Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Case Photovoltaic power generation project at the Second Chinese Medicine Factory Energy and Resource Management
To reduce energy consumption intensity and GHG
emissions during operations the Company advanced
the construction of a photovoltaic power generation Energy Utilization
project at its Second Chinese Medicine Factory and
connected it to the grid. Since its commissioning the
project has operated stably overall and the substitution In accordance with the Energy Law of the People's Republic of China the Company has formulated and issued the Hapharm Group
effect of clean energy has gradually become apparent. Energy Management Measures establishing a standardized energy management system. On this basis the Company coordinates
During the reporting period the photovoltaic system its subsidiaries to advance energy management work implements full-process supervision inspection and assessment of their
generated a total of 500200 kWh of electricity provid- performance guides special actions for lean energy conservation and strengthens key matters such as internal audits of energy
ing practical support for the Company's efforts to management.address climate change and optimize its energy Photovoltaic panels at the Second
structure. Chinese Medicine Factory
Each subsidiary of the Company has formulated energy management systems
which include content related to energy procurement use conservation and
Impact Risk and Opportunity Management evaluation.The Company integrates climate change-related impacts risks and opportunities into a unified environmental and operational
management framework continuously conducting systematic identification and dynamic management. Through energy use analy- The Company establishes energy consumption ledgers and conducts energy use
sis and production operation assessments the Company identifies the potential impacts of factors such as extreme weather analysis. The Company records and tabulates actual energy consumption daily
temperature changes policy tightening and technological iteration on production operations cost structure and supply chain performs summary analysis periodically to promptly identify anomalies in energy
stability and conducts comprehensive evaluations based on the degree of impact and frequency of occurrence. Based on the consumption finds the causes and formulates solutions.evaluation results the Company continuously optimizes its energy conservation and carbon reduction management measures
integrates relevant requirements into production operations and management decision-making processes and strengthens
continuous control over climate-related risks through data monitoring and capacity building. While effectively preventing environ-
mental and compliance risks the Company also focuses on new health demands and market expansion opportunities brought by The Company strengthens management and control by establishing energy
climate change promoting synergy between environmental management and business development. consumption quotas for production lines. Data on energy consumption for more
than ten eligible production lines is accumulated quotas are established and
these quotas are continuously optimized based on actual usage.Indicators and Objectives
At the current stage the Company's carbon management focuses on Scope 1 and Scope 2 emissions and is steadily incorporating The Company enhances monitoring of high-consumption equipment. Metering is
Scope 3 emissions into its control system. It has already initiated the identification of core emission sources specifically covering added to some high-energy-consuming equipment a monitoring system is estab-
carbon emissions generated from employee commuting business travel the production of purchased raw materials and product lished data on equipment energy consumption is collected energy use trends are
transportation processes. analyzed and operating statuses are reasonably adjusted to ensure efficient
operation.Indicator name1 Unit 2025 data
Scope 1 greenhouse gas emissions Tonnes of CO equivalent ?????.?? 1.The Company strengthens energy use inspections to reduce energy waste. Daily 2
inspections are conducted on the energy preparation transmission and distribu-
tion systems to prevent leaks drips and spills thereby reducing energy waste.Scope 2 greenhouse gas emissions Tonnes of CO2 equivalent ??????.??
Total greenhouse gas emissions Tonnes of CO2 equivalent ??????.?? The Company's energy use management measures
1The Company calculates its greenhouse gas emissions mainly in accordance with relevant standards and methodologies of the GHG Protocol and conducts
measurement in combination with relevant national and industrial regulations.
71 72About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Case Strengthening internal energy audit and control to solidify the foundation for energy conservation Case Upgrading the automatic control of the heating system to reduce steam consumption and and cost reduction increase efficiency
In 2025 the Company systematically conducted The original heating systems in some workshops
internal energy audits on-site special inspections of Harbin Pharmaceutical General Factory were
and energy conservation awareness campaigns for incapable of temperature adjustment. Constant
its subsidiary production enterprises. Weaknesses heating was still maintained during non-produc-
in energy management were identified and tion hours at night resulting in energy waste. In
addressed resulting in a total of 35 energy-saving 2025 the factory installed a new automatic
improvement recommendations being issued. In control system which can automatically adjust
October of the same year the Company held a heating parameters according to preset tempera-
special meeting on energy management. Through a tures and time periods thus effectively improving
systematic review of the effectiveness and short- energy utilization efficiency. According to estima-
comings of the internal audit work and specialized tion the renovation will reduce annual heating Addition of an automatic control system for the
training sharing practical ideas for energy-saving heat exchange stationsteam consumption by approximately 540 tons.renovation projects the Company comprehensively Energy internal audit training
enhanced its level of refined energy management
helping to achieve the strategic goals of energy
conservation cost reduction and efficiency
improvement.Green Office
The Company continuously promotes green office management in its daily operations. By advocating paperless offices online
Case Upgrading drying equipment heaters to achieve dual improvements in energy efficiency and production capacity meetings conservation of electricity and water and recycling of office supplies it reduces resource consumption and unneces-
sary energy waste. At the same time considering the operational characteristics of its office spaces the Company explores the
In January 2025 Shiyitang Chinese Medicine application of energy-saving technologies in office buildings while ensuring a comfortable and stable working environment gradu-
completed the upgrade and renovation of its fluid ally improving the energy utilization efficiency of its office operations.bed dryer heater. Following the renovation the
drying efficiency of the equipment was significantly
improved; the single-batch operating time of the
fluid bed dryer in the drug granulation process is
expected to be reduced by two hours achieving the
Case Green operation practices for office buildingsdual goals of enhancing energy efficiency and
increasing production efficiency. This renovation
effectively improved equipment operating efficiency To respond to national energy conservation and emission reduc-
and reduced production energy consumption. tion policies and improve the heating efficiency of its office
During the reporting period it saved a cumulative building the Company implemented a smart heating energy-sav-
Upgrading and renovation of the fluid bed dryer heater
total of 23000 kWh of electricity and 76.8 tonnes ing renovation project piloting it at its headquarters office
of steam accumulating practical experience for the building and associated heat exchange station. The project
Company's future energy-saving optimizations in achieved refined management and dynamic adjustment of heating
production processes. operations through measures such as independent operation
renovation of the heating system optimization of the secondary
pipe network online monitoring of indoor temperatures and
upgrading of heat metering methods. This effectively improved
heating quality and avoided energy waste. Since the project's
completion and stable operation it has saved approximately 150
tonnes of standard coal annually reduced carbon dioxide
emissions by about 540 tonnes and simultaneously lowered
emissions of nitrogen oxides sulfur dioxide and particulate Smart heating energy-saving
matter playing a positive role in mitigating climate change and renovation project
improving regional environmental quality.
73 74About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Case Practicing the concept of green travel and deepening energy conservation and Water Resource Utilizationemission reduction practices
In January 2025 in response to the national policy orientation of The Company strictly adheres to relevant national and local laws regulations and policies systematically promoting the refined
energy conservation and emission reduction and in light of the management of water resources. Upholding the principles of scientific management and continuous improvement the Company
operational demands of cost reduction and efficiency improve- focuses on the core goals of efficient and intensive use of water resources cost reduction and efficiency improvement. By
ment the Company officially launched the special program for the strengthening control measures throughout the entire water usage process it achieves the dual effects of resource protection and
new energy replacement of official vehicles. The Company waste elimination.replaced 11 fuel vehicles approaching the end of their service life
under itself and 9 affiliated enterprises with domestic hybrid The Company strengthens the daily operation maintenance and control of water-using equipment conducting targeted inspec-
models. The carbon emission per vehicle decreased by approxi- tions and rectifying problems to reduce water resource waste from leaks drips and spills. In its production and operation process-
mately 40% year-on-year which effectively facilitated the enter- The Company's new energy official vehicles es the Company utilizes a circulating water system achieving the dual goals of reducing water consumption and improving utiliza-
prise’s green and low-carbon transformation process and tion efficiency through optimized water resource allocation. Additionally on the annual environmental protection and water
accumulated replicable and generalizable practical experience for conservation day the Company conducts water-saving publicity and education in various forms to raise employee awareness
the Company to achieve its dual carbon goals. laying a solid foundation for building a water-saving enterprise and promoting corporate sustainability.Key performance
Key performance
Category Indicator Unit 2025 data
Indicator name Unit 2025 data
Direct energy consumption Tonnes of standard coal 15778.58
Total water consumption Tonnes 6320252
Gasoline consumption Tonnes 116.71
Diesel consumption Tonnes 84.84 Freshwater consumption Tonnes 1074961
Natural Gas Consumption 10 Thousand Cubic Meters 1164.15
Recycled water consumption Tonnes 11454932
Energy Indirect energy consumption Tonnes of standard coal 31201.88
usage Water consumption intensity
per million in revenue Tonnes/RMB 1 million in revenue 395.91
Purchased electricity MWh 58192.01
Purchased Steam GJ 704867.68
Total energy consumption Tonnes of standard coal 46980.46
Comprehensive energy consumption per Tonnes of standard
unit of revenue coal/million 2.94
Total clean energy consumption Tonnes of standard
(photovoltaic natural gas) coal 16070.24
Clean Solar energy consumption (photovoltaic) MWh 4784.65
energy
Proportion of clean energy consumption to total energy
consumption % 34.21
75 76About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Product System for Value
Management Special Topic to Solidify a Foundation for Advance Low-Carbon Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation
to Build a Safe and
Management Shared Future
Pollutant Emissions Exhaust Gas Management
As a pharmaceutical enterprise committed to environmental responsibility the Company strictly complies with core regulations
Wastewater Management such as the Environmental Protection Law of the People's Republic of China and the Air Pollution Prevention and Control Law of
the People's Republic of China systematically advancing waste gas management. The Company's waste gas primarily consists of
process emissions originating from basic production steps such as crushing and packaging as well as core processes including
The Company strictly adheres to the requirements of laws and regulations such as the Environmental Protection Law of the active pharmaceutical ingredient synthesis and solvent recovery. Its characteristic pollutants are mainly particulate matter and
People's Republic of China and the Water Pollution Prevention and Control Law of the People's Republic of China to carry out volatile organic compounds.full-process wastewater management. Each subsidiary is equipped with a standardized wastewater treatment station and profes-
sional technicians dedicated to process parameter control and system operation management ensuring that treated wastewater
meets discharge standards consistently. The management of production wastewater fully implements the core control principle of
"separating clean water from wastewater treating wastewater based on its quality and ensuring compliant discharge." Key performance
Indicator name Unit 2025 data
Case Building a standardized sewage station to control emissions and treat pollution
Total waste gas Cubic meter 21560.51
Sanchine has constructed its own wastewater treatment station with
a daily capacity of 3200 tonnes utilizing the sequencing batch
reactor (SBR) activated sludge process for wastewater pretreatment. Particulate matter (PM) Tonnes 2.55
The treated effluent quality consistently meets the requirements of
the Discharge Standard of Water Pollutants for Pharmaceutical
Industry Mixing/Compounding and Formulation Category before Sulfur oxides (SOx) Tonnes 0.48
being discharged into municipal sewage treatment plants via the
municipal pipe network. Sanchine strictly adheres to technical speci- Nitrogen oxides (NOx) Tonnes 18.82
fications in configuring and upgrading online wastewater monitoring
facilities while also commissioning third-party professional monitor-
ing agencies to conduct regular testing of wastewater pollutants at Wastewater treatment station Volatile organic compounds (VOCs) Tonnes 172.61
the plant boundary. By real-time monitoring of discharge data
precise control of pollution control facility operations and continu-
ous optimization of treatment processes the Company enhances the
effectiveness of its pollution control efforts.Noise Management
Key performance
The Company strictly adheres to the relevant requirements of the Law of the People's Republic of China on the Prevention and
Control of Noise Pollution to implement noise control measures upholding the governance principle of "source control and
Indicator name Unit 2025 data transmission path sound insulation." In equipment selection priority is given to low-noise facilities to minimize noise generation
at the source. For high-noise equipment such as air compressors and fans independent soundproof rooms are provided. Produc-
Total amount of wastewater discharge Tonnes 1052611 tion pipelines are treated with flexible connections and sound-insulating wrapping to reduce noise transmission. Additionally
green belts are planted along the plant boundaries to create natural noise barriers. Through these targeted control measures the
Company effectively implements noise governance requirements ensuring harmonious coexistence between the production
Chemical oxygen demand (COD) Tonnes 38.86 environment and the surrounding area.Ammonia nitrogen (NH?-N) Tonnes 1.28
77 78About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Waste Management Classified Standardized Compliant
storage transportation disposal
The Company strictly complies with regulations such as the Law of the People's Republic of China on the Prevention and Control
of Environmental Pollution by Solid Waste and the Regulations on the Management of Municipal Solid Waste to carry out waste 1 2 3
management. Aligning with the characteristics of the pharmaceutical sector it also implements the requirements of the Measures Strict implementation Internal transporta- Recyclable waste is
for the Administration of Hazardous Waste Transfer. Guided by the core policy of "reduction resource recovery and harmless of zoned and tion utilizes dedicat- processed by
disposal" the Company has established a tiered management system encompassing "source reduction—recycling—end-of-pipe classified storage ed sealed containers compliant renewable
harmless disposal" and implemented a standardized closed-loop process of "classified storage—standardized transportation— management and transport resource units while
compliant disposal." specifications is vehicles while non-recyclable
carried out fulfilling external transporta- hazardous waste is
the "four prevention" tion of hazardous disposed of
Reduction requirements of
measures By optimizing processes to improve packaging efficiency and reduce material loss the generation of
waste is entirely harmlessly in
anti-seepage
waste and discarded packaging materials is minimized at the source. entrusted to units accordance with laws
anti-loss anti-disper- possessing road and regulations.sion and rain transport permits and
protection while hazardous waste
simultaneously disposal qualifica-
Resource equipping emergency tions;
recovery and material reserves;
utilization For high-concentration waste solvents priority is given to recovery and purification through processes
such as distillation and rectification followed by reuse in production processes to reduce hazardous
waste generation. The Company's waste disposal process
General Waste Management
Harmless
disposal Non-recyclable hazardous waste is managed by category according to its hazardous characteristics
and entrusted to qualified professional institutions for harmless disposal. The Company's general waste mainly includes domestic waste generated by employees office paper waste used packaging
materials and food waste. The Company implements classified collection and standardized storage for general waste entrusting
legally qualified third-party units for removal and disposal to ensure that the disposal process is compliant safe and orderly. At
the same time by promoting continuous flow production and optimizing production organization methods the generation of
The Company's waste management policy
general waste and pollutants is reduced.Hazardous and Medical Waste Management
The Company's hazardous waste mainly includes laboratory waste liquid waste ion exchange resins non-conforming products
and laboratory waste. All types of hazardous waste are strictly managed by category in accordance with the National Catalogue of
Hazardous Wastes and incorporated into the unified hazardous waste management system according to pharmaceutical industry
hazard categories (e.g. HW02 HW49).The Company implements the principle of "strict management" for hazardous waste clarifying the responsibility requirement of
"who pollutes who treats." Through systems such as the Environmental Protection Management Measures Hazardous Waste
Management Measures and Emergency Response Plan for Environmental Emergencies the Company standardizes the manage-
ment of the entire process of hazardous waste generation collection storage transfer and disposal.
79 80About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Specific operational procedures include: Key performance
Hazardous waste is centrally collected in the production system and quality inspection workshops and then transferred to
the temporary hazardous waste storage room;
The temporary storage room is constructed in accordance with the Standard for Pollution Control on Hazardous Waste Indicator name Unit 2025 data
Storage and implements classified management with clear and conspicuous hazard signage;
For off-site disposal all hazardous waste is entrusted to professional units holding a Hazardous Waste Operation License Total hazardous waste Tonnes 210.46
to complete reception and compliant disposal within a fixed timeframe.Hazardous waste intensity Tonnes/RMB 1 million in revenue 0.01
Total non-hazardous waste Tonnes 2075.38
To ensure employee safety the Company requires strict adherence to personal protective equipment requirements during the Non-hazardous waste intensity Tonnes/RMB 1 million in revenue 0.13
sorting transfer and temporary storage of hazardous waste. Additionally designated positions fixed schedules and defined
routes are implemented to minimize secondary pollution and safety risks.Waste recycling rate % 91.42
During the reporting period the Company
During the reporting period ???% of the waste generated by the Company was disposed of in compliance
with regulations with no incidents of illegal discharge or transfer occurring. Ecosystem and Biodiversity Conservation
The Company places high importance on the interrelationship between raw material sources production activities and the natural
ecological environment. While complying with national laws and regulations on ecological and environmental protection the
Company continuously identifies potential impacts of its business activities on ecosystems and biological resources. It prudently
advances risk prevention control and management related to biodiversity striving to reduce adverse environmental impacts while
ensuring stable production and operation.Case Closed-loop reclamation of organic solvents to improve quality and efficiency in resource utilization
The Company integrates ecological and environmental protection concepts into its planting and management processes. By
In the synthesis workshop organic solvents are prioritized for regularly monitoring and assessing soil conditions in planting areas and tracking changes in soil physicochemical indicators the
cooling circulation and reuse within the production process. Company prevents potential impacts on soil ecology caused by improper planting or input usage promoting the coordinated
When concentrations fall below usage standards they are development of Chinese materia medica production and the local ecological environment.transported via pipelines to the solvent reclamation workshop
for purification before being returned for reuse. The solvent
reclamation workshop primarily handles the reclamation of
various organic solvents from the synthesis workshop and the
sterile API workshop with main products being reclaimed
anhydrous ethanol and industrial ethanol. For other organic
solvents reclaimed from the synthesis workshop the Sanjing
Mingshui workshop periodically conducts negative pressure
distillation and reclamation in a multi-functional tower. Qualified
products are sent to finished product tanks while non-conform-
ing products are temporarily stored in intermediate tanks for
Solvent reclamation workshop
reprocessing. This entire process maximizes resource utiliza-
tion minimizes losses and ensures the quality of reclaimed
products. Throughout the year a total of 50 tonnes of ethanol
and 10 tonnes of ethylene glycol were saved.
818204
Employee Development
to Build a Safe and
Shared Future
The content of this chapter
Employee Rights and Care ??
Talent Cultivation and Development ??
Occupational Health and Safety ??
Rural Revitalization Practices ???
Public Welfare and Social Action ???About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon Creation to Build a Safe and
Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Management Shared Future
Opportunity type Specific description Financial impact Response measures
Employee Risks and Opportunities Time horizon
A comprehensive human resource Continuously optimize the
management system has been human resource manage-
established. Through measures such ment system improve
Focusing on employee team stability and organizational sustainability goals the Company systematically identifies and assesses as strengthening institutional career paths build a
employee-related risks and opportunities integrating management requirements into human resources management and daily guarantees deepening training and full-cycle training system
operational processes. In conjunction with business development plans and the actual workforce situation the Company continu- development and optimizing Talent Medium and and a fair and reasonable
compensation and incentives the
ously evaluates the potential impacts of factors such as talent mobility person-job fit and occupational health implementing retention long term compensation system
Company aims to achieve sustainable
dynamic monitoring and response through existing management mechanisms. empower sustainable talent
talent career development reduce the development and solidify
turnover rate of core talent and the foundation for talent
ensure the inheritance of organiza-
Reduced retention.Risk type Specific description Time horizon Financial impact Response measures tional experience and the continuous Employ- stable development of the business. recruitment
ment and training Strengthen promotion and Deepen school-enter-
costs and publicity of the employer
Influenced by objective factors such as prise cooperation to oppor-
tunities improved labor brand fully showcase the Harbin's geographical location and broaden channels for Through standardized employment efficiency Company's compliant
regional economic development levels talent attraction and
Rising labor practices and caring initiatives the employment practices and
Regional some high-quality talent exhibits lower cultivation; strengthen Employer
Employ- costs and Company continuously enhances its employee care initiatives
recruitment job-seeking willingness leading to Short and employer brand building brand Medium and
ment declining employer brand influence and market actively participate in
disadvantag- certain recruitment pressures for the medium term by highlighting the enhance- long term
risks workforce reputation attracting high-quality industry employer brand
es Company and constraining the Company's develop- ment
productivity talent and creating a positive talent awards and continuously
efficiency and progress of personnel ment platform and siphon effect. enhance brand influence
replenishment. career stability to and reputation.continuously enhance
talent attraction. Open up professional Clear and smooth career development
promotion channels for
Examples include fire and paths are established and diversified R&D personnel establish
explosion risks associated with incentive mechanisms are improved to
Fire and innovation incentives and Short and
chemicals as well as fire hazards Stimula- fully stimulate employee innovative Revenue growth
explosion Talent special awards encourage medium term
arising from the indoor storage of tion of vitality and creative enthusiasm. Medium and and enhanced cultiva- employees to participate in
diesel generators. innovative Technological breakthroughs and long term workforce tion challenging projects and
vitality management innovation are promoted productivity
cross-departmental
Increased asset Strengthen control to continuously enhance the collaboration and foster a
losses production over fire safety Company's core competitiveness.Examples include distribution boxes positive and upward-look-
stoppage losses mechanical
Safety Electric in the water supply pump room of Short and ing innovation culture.compensation equipment safety
risks shock the power workshop which may medium term expenses and devices and Implement annual
cause electric shock injuries.emergency personnel safety employee physical
response costs behaviors. The working environment and examinations and
occupational health management are occupational health
continuously optimized to effectively monitoring optimize labor
Health Employee
protect the physical and mental health Medium and Lower medical protection equipment and
Examples include risks of mechani- protec- health and absentee-
Mechanical Medium and of employees reduce sick leave long term establish caring facilities
cal injury in production packaging tion protection
injury long term absentee rates and further enhance
ism costs such as mother-and-baby
and maintenance positions. employee loyalty and work engage- rooms and fitness activity
ment. areas to promote humanis-
tic care-based safety
management.Dust and Dust hazards may be generated
Short and
chemical during sub-packaging in
medium term Higher medical Strengthen control Occu- hazards production workshops. expenses reduced over equipment and
pational
workforce produc- facility sealing and
health
tivity and increased personnel protection
risks Noise hazards may be generated
Short and related management.Noise by air conditioning systems in
medium term
production workshops.
85 86About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
The Company has established a comprehensive compensation and performance grievance mechanism clearly defining channels
Employee Rights and Care procedures and time limits to protect the legitimate rights and interests of employees. Regarding compensation employees may
file a grievance within 30 days of the salary payment date if they have objections. For performance employees may submit a
grievance application to the Performance Management Office within five working days after the announcement of assessment
results which will investigate and rule accordingly. Grievance channels include offline feedback email systems and union
The Company strictly complies with national laws and regulations such as the Labour Law of the People's Republic of China and channels ensuring that employee concerns are handled in a timely and fair manner.the Labor Contract Law of the People's Republic of China adhering to the principles of legal employment and standardized
management and resolutely prohibits the use of child labor and forced labor. The Company has established a comprehensive
human resource management system including the Internal Competition Management Measures Compensation Management Employees may typically exercise their right to appeal through channels such as offline feedback email
Measures Employee Transfer Management Measures and Employee Reward and Punishment Management Regulations to systems and cloud mailboxes in accordance with regulations. Offline feedback is generally directed directly
effectively protect the legitimate rights and interests of employees and firmly prevent all types of illegal and non-compliant Adminis- to the Human Resources Department which investigates handles and responds. If there are objections to
employment practices. trative the offline handling results further appeals may be submitted via the Company email system or cloud
channels mailbox. The cloud mailbox is managed and supervised by the President's Office with handling results and
The Company has established a diversified and standardized recruitment process to attract outstanding talent from various feedback reported to company management to ensure timely and effective resolution of employee appeals.backgrounds. It fully considers the diverse backgrounds of candidates using abilities experience and skills as core evaluation
criteria during selection eliminating all forms of discrimination based on gender age marital status etc. to ensure fairness and
impartiality in recruitment. The Company strictly prohibits inappropriate behaviors such as verbal abuse and harassment and
imposes corresponding penalties based on the severity of violations aiming to create a positive working atmosphere for employ-
ees. Trade Employees may file complaints with the trade union. If the trade union verifies that the Company has
union infringed upon employees' labor rights and interests it has the right to legally demand corrections and
The Company continuously optimizes its recruitment management system establishing a channel strategy of "internal recruitment channels assume corresponding responsibilities.as the mainstay and external recruitment as supplementary." Talent quality assessment standards have been set screening
resumes based on factors such as age education corporate system experience and career stability. Interviews are evaluated
across four dimensions: company benchmarking competency benchmarking motivation benchmarking and cultural benchmark-
ing ensuring strict control over talent quality. Campus recruitment quality is rigorously controlled by assessing education institu-
In accordance with government requirements the Company has established a dedicated department for
tion major and political affiliation. Background checks conducted through third-party agencies mitigate hiring risks at the source
employee rights protection. Employee appeals can be accepted and filed through this department which
comprehensively improving recruitment quality. In 2025 the Company recruited 794 external talents including 115 management Other
urges relevant responsible departments to issue responses in accordance with regulations and facilitates
trainees achieving a notable improvement in talent quality. Among them 68.7% were from top-tier universities (985/211) repre- channels
review by superior competent authorities ensuring that reasonable employee demands are properly
senting a year-on-year increase of 32.1%; 61.7% held master's or doctoral degrees a year-on-year increase of 16.2%; and 29.6%
resolved.were Party members a year-on-year increase of 13.2%. The overall talent structure continued to be optimized.Employee compensation and performance grievance channels
Compensation and Performance Management
In democratic management the Company widely solicits employee opinions through channels such as the Employee Representa-
tive Congress and employee representative proposals. It carries out democratic management practices that are rich in content
distinctive in character and diverse in form effectively safeguarding employees' rights to know participate and supervise.In terms of leave and compensation management the Company strictly complies with laws and regulations such as the Regula-
tions on Paid Annual Leave for Employees and the Measures for the Implementation of Paid Annual Leave for Enterprise Employ-
ees ensuring that employee working hours meet legal requirements and safeguarding their right to rest and take leave. The
Company has formulated a unified compensation management system to standardize salary distribution and incentive manage-
ment for headquarters and subsidiaries. It has optimized the compensation structure by implementing a "broadband compensa-
tion" model reinforcing the incentive orientation of "paying for position value and paying for ability and performance." The Compa-
ny scientifically conducts job value assessments optimizes the job value map and clarifies career development paths for employ-
ees in both management and professional sequences. Within the salary bands employee compensation is adjusted based on
comprehensive performance evaluation results fully reflecting differences in employee capabilities and performance contribu-
tions thereby building a fair and reasonable compensation system.Employee Care Activities
87 88About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Each year the Company conducts an administrative service
Care and Benefits satisfaction survey for all employees. The questionnaire is
designed around seven key dimensions: property manage-
ment catering sanitation meetings shuttle buses official
In terms of employee care the Company has developed diverse heart- vehicles and security. The effective response rate reached
warming initiatives to meet the differentiated needs of different groups. 98% and the average employee satisfaction with these
For female employees a series of activities was organized for Interna- service areas reached 93% reflecting the Company's contin-
tional Women's Day including a corporate culture knowledge quiz a uous optimization of administrative service quality.female employee photo contest a visit to the Hayaoliu Print Museum
distribution of product discount coupons and a "Rose and Book The Company actively carries out a promotion month for
Fragrance" reading event. "Lilac" demonstration classes for female factory affairs transparency and democratic management
employees covering embroidery tie-dye and gourd pyrography were publicizing related knowledge through forms such as knowl-
opened to enrich their spiritual and cultural lives. Additionally edge competitions and employee representative training
mother-and-baby rooms were constructed to provide comfortable resting thereby strengthening the foundation for democratic Staff Team Building Activities
and nursing spaces for breastfeeding female employees. For employees management.facing difficulties regular support activities such as holiday visits Outstanding Female Workers Award in
HPGF Safety Training
assistance for major illnesses and care for children are carried out to
convey corporate warmth and enhance employees' sense of belonging.The Company has established a comprehensive employee benefit system Key performance
covering statutory benefits and company benefits. Statutory benefits are
strictly implemented in compliance with laws and regulations including
social insurance housing provident fund and other statutory welfare Indicator name Unit 2025 data
policies forming a solid baseline for protecting employee rights. Compa-
ny benefits encompass three categories: basic protection special Total number of employees Persons 10039
supplements and special care. In 2025 the Company further deepened
By gender
benefit practices by reaching group purchasing agreements with external
partners adding high-quality and cost-effective services such as refuel- Number of female employees Persons 4922
ing and insurance enriching benefit offerings and effectively enhancing
HPS Women's Day Activities
employees' sense of gain. Number of male employees Persons 5117
By age
Benefit type Content covered Number of employees aged 30 and below Persons 995
Number of employees aged 31 to 40 Persons 3427
Social insurance and housing provident fund: In compliance with laws and regulations pension Number of employees aged 41 to 50 Persons 3926
insurance medical insurance (including maternity insurance) unemployment insurance work-related
injury or ill health insurance and housing provident fund are paid for employees and relevant Number of employees aged 51 and above Persons 1691
Statutory benefits regulations of the state and local governments where the Company and its subsidiaries are located
regarding social insurance and housing provident fund are strictly implemented. Number of newly hired employees Persons 794
Benefit policies: National and local employee benefit policies are fully implemented and relevant Number of newly hired female employees Persons 428
requirements such as employee leave and benefits for single-child families are strictly enforced.Number of ethnic minority employees Persons 393
Number of employees with disabilities Persons 160
Basic benefits: These include in-kind holiday benefits annual physical examinations commuting
support free lunches and employee team-building activities. Employee turnover rate % 6
Special benefits: Based on the Company's actual situation and job characteristics supplementary Employee benefit coverage rate % 100
Company benefits insurance rental subsidies for off-site work tea break snacks fitness and entertainment facilities
and low-price group purchases are provided. Employee satisfaction survey coverage rate % 100
Special care benefits: Exclusive benefits such as mother-and-baby rooms are established to meet Employee trade union coverage rate % 100
the needs of specific employee groups.
89 90About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Product System for Value
Management Special Topic to Solidify a Foundation for Advance Low-Carbon Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation
to Build a Safe and
Management Shared Future
Talent Cultivation and Development Talent Development System
To achieve "standardization process-orientation institutionalization informatization digital intelligence and assessment-based The Company has constructed a talent development system centered around the three dimensions of "schools instructors and
management" the Company is comprehensively promoting the optimization and upgrading of its human resources system. This courses." It has refined the course matrix for all job categories and levels focusing on core areas that directly impact business
includes implementing "full life-cycle" career management for employees and building a human resources management system that operations such as marketing market development e-commerce and R&D. Key emphasis is placed on cultivating critical groups
aligns with market principles and matches the corporate culture. The Company focuses on building an incentive-oriented compensa- including management trainees reserve cadres and high-potential employees. The Company fosters an organizational
tion and performance system and a capability-oriented talent development system. It strengthens the development of its cadre team atmosphere combining "learning practicing testing and competing" forging practical skills through project immersion and job
and talent echelon adhering to the principle of "promoting the capable and demoting the mediocre." The Company increases invest- rotation ensuring that talent growth resonates with business development.ment in talent cultivation empowering management trainees and young cadres to grow through practical experience thereby stimu-
lating organizational vitality and providing talent assurance for the implementation of the Company's strategy and high-quality devel- The Company possesses a comprehensive position management and talent development system providing clear and well-defined
opment. career advancement channels for all job sequences including R&D production marketing communication and functional roles.In terms of talent development the Company has assembled a professional faculty composed of department leaders and business
backbones built a customized curriculum system and adopted diverse learning formats such as sharing sessions attendance at
System and Framework Setup important meetings work reports and study tours. For management personnel echelon development is strengthened through job rotation work reviews and case studies. For grassroots personnel the "master-apprentice" mentorship mechanism is continuous-
ly promoted. Concurrently a dynamic talent reserve pool has been established to provide solid support for both talent growth and
corporate development. In 2025 the Company was recognized as a Harbin Municipal High-Skilled Talent Training Base and
received the "AI Application Pioneer Award" from Beijing Beisen Cloud Computing Co. Ltd.The Company has formulated the Position System Management Measures to standardize position information and job responsibili-
ties laying a foundation for talent development. It has also formulated the Talent Development Management Measures which
based on knowledge skills and experience requirements for positions constructs diverse development paths that vertically cover
management professional and operational tracks and horizontally support cross-track transfers. The R&D System Professional
Track Qualification Rating Scheme (Trial) has been formulated to detail professional promotion standards for R&D personnel Case Digitalization empowering internal learning and capability enhancement
opening up a seven-level professional development channel from assistant researcher to chief scientist providing clear guidance
for R&D talent growth. At the same time the Company has formulated and implemented the R&D Project Incentive Scheme (Trial)
centered on full life-cycle management of R&D projects scientifically setting key milestone nodes and assessment indicators to In April 2025 the Company introduced an intelligent learning
continuously stimulate the innovative vitality of the R&D team. platform software to host internal and external learning
resources and shorten course production cycles using
AI-assisted course creation. Leveraging AI course creation
The Company has established and improved a talent development system clarifying career development paths and promotion
features the Company developed a total of 206 AI courses
rules through multiple policies. It also implements job value assessments comprehensively evaluating job value based on seven
during the reporting period saving approximately 500 hours
factors: span of control scope of responsibilities organizational impact environmental conditions problem-solving capabilities
in course recording and post-production time effectively
qualifications and communication requirements. In 2025 the Company further optimized its job mapping scientifically differenti-
enhancing the efficiency of training resource development.ated relative differences in job values and refined the position management system. The Company formulated the Performance
Based on business scenarios Renmintongtai applied AI-pow-
Management Measures to implement closed-loop performance management adhering to the principle of "comprehensive horizon-
ered practice partner features to conduct sales staff training
tal coverage and thorough vertical implementation." This ensures that all employees sign performance contracts and strengthens
focusing on strengthening pre-sales recommendations and
the application of performance results. Assessment outcomes are linked to performance bonuses merit-based salary adjust-
drug follow-up capabilities. This not only improved training AI Application Pioneer Award
ments cadre reappointment competitive promotion reserve talent selection and targeted training thereby motivating employees
efficiency but also facilitated the conversion of training
to achieve high performance. The Company implements the Cadre Appointment Management Measures following the principle of
outcomes into actual business performance.integrity and ability and selects cadres through a combination of open competition and organizational appointment. Additionally
the Company enforces a "one-year appointment" system for management personnel. Through comprehensive evaluation of perfor-
mance assessments competency appraisals work reports and daily rewards and punishments the Company determines eligibili-
Adhering to the talent attraction philosophy of "gathering
ty for cadre reappointment continuously building a high-quality and professional management team.people with brand retaining them with ecosystem motivat-
ing them with value warming them with policy and nurtur-
ing their growth" the Company expanded its employer
brand influence through 12 campus presentations 23
dual-selection fairs and university-enterprise collabora-
tions. It established a "Talent Subsidy" to enhance talent
attraction and implemented the "Harbin Talents New Policy
30 Articles." By leveraging competitive salaries and local Shenyang Pharmaceutical Dongbei University of Finance
policies the Company empowers the localized development University Job Fair and Economics Job Fair
of talent achieving win-win growth for talent enterprise
and city.
91 92About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation
to Build a Safe and
Management Shared FutureCase “Voyager Training Camp" program for reserve cadres Case University-enterprise collaboration: Advancing the joint cultivation mechanism for TCM talentTo support the Company's strategy and talent needs and strengthen the reserve cadre echelon the Company The Company continuously improves its diversified talent development system to meet the needs of the traditional
launched the first "Voyager Training Camp" for reserve cadres from March to October 2025 in accordance with Chinese medicine (TCM) industry. During the reporting period Shiyitang Chinese Medicine signed a joint cultiva-
the Reserve Cadre Selection and Development Plan. The program was held from March to October 2025 at tion agreement with the School of Pharmacy Heilongjiang University of Chinese Medicine and co-established a
Heilongjiang University and the Headquarters Multi-function Hall. Training content covered diverse courses joint cultivation base promoting the implementation of a university-enterprise collaborative education mechanism.including leadership enhancement management innovation marketing and finance. Teaching methods included By co-building practical training platforms involving corporate mentors in teaching and organizing students to
classroom interaction case studies and project presentations to systematically improve the comprehensive participate in actual R&D and production projects the two parties improved the alignment between talent cultiva-
abilities of reserve cadres. A total of 76 trainees participated in this session; currently 13 have been successfully tion and industry needs enhancing students' practical skills and professional identity. Simultaneously leveraging
promoted representing a promotion rate of 17% securing a pool of high-quality management talent for the the resource advantages of both the university and the enterprise in research and industry they engaged in
Company's development. exchange and cooperation in areas such as TCM R&D and the development of "Cold-Region Longjiang Medicines"
resources. This explores a talent development path connecting teaching practice and scientific research provid-
ing stable talent support for the Company's sustainable development and the high-quality development of the TCM
industry.Leadership enhancement and management Financial management training course
innovation training course
Case "Chunlei Training Camp" for the 2025 Cohort of Management Trainees
Joint cultivation by Shiyitang Chinese Medicine and
Heilongjiang University of Chinese Medicine
In July 2025 the Company launched the "Chunlei Training Camp" for the 2025 cohort of management trainees.Themed "Hapharm Group's New Stars: Chasing Dreams Forward Daring to Compete and Win" the program provid-
ed 115 management trainees with a 10-day immersive intensive training. The training featured systematic courses
including team-building exercises corporate culture strategic branding marketing planning and professional Key performance
ethics. Through diverse teaching formats such as centralized instruction site visits and study tours senior execu-
tive symposiums and project discussions and presentations the program comprehensively empowered the
growth of management trainees. It efficiently bridged the "last mile" from campus to the workplace helping new Indicator name Unit 2025 data
employees quickly integrate into the enterprise and become competent in their roles.Total training hours Hours 248712.2
Average training hours per person Hours 24.77
Total number of training sessions for the year Times 1968
Total investment in employee training Ten thousand yuan 295.71
Number of employee training participants Persons 9869
Employee training coverage rate % 98
Systematic curriculum training session Team-building exercises Percentage of employees receiving regular
performance and career development reviews % 100
93 94About This Message About 2025 Key Hapharm Performance in Sustainability
Governance and Compliance Green Operations to Employee Development
Report from the Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
In recent years the honors and awards received by Hapharm Group and its subsidiaries are as follows:
Occupational Health and Safety
The Company has constructed a comprehensive safety management system. A Work Safety Committee is established as the
highest authority for safety management responsible for decision-making on major work safety matters providing business
support and formulating overall safety management policies. A dedicated safety management department the EHS and Engineer-
ing Equipment Department is responsible for setting safety assessment goals and indicators establishing and operating the Hayao General Factory Sanchine
safety management system conducting performance assessments and investigating and handling major safety accidents. All
subordinate industrial R&D and commercial units have established safety management departments responsible for decompos- In 2024 it was awarded the In 2023 it was awarded the
ing and implementing safety management goals and indicators building and ensuring compliant operation of systems pursuing title of Advanced Organization title of Winning Unit in the
continuous improvement providing business training fostering a safety culture and investigating and handling safety accidents Unit in the Harbin Ankang Cup 2021-2022 Heilongjiang
within their respective units. Competition and the Province Ankang Cup
recycling group of Workshop in 2024 it was awarded the
301 was awarded the title of title of Winning Unit in the
Excellent Team in the Ankang 2022-2023 Harbin "Ankang
Cup. Cup" Competition.President
EHS and Engineering Equipment Department
Shiyitang Chinese Medicine Sanjing Mingshui
In 2024 it won the "First Prize in In 2024 it was awarded the title
the Team Competition of Mini of "2024 Suihua City Occupa-
Fire Stations for Key Fire Safety tional Health Enterprise."
Units in Daoli District."
Organizational structure chart of the safety management
system of Hapharm Group
The Company has formulated systems such as the Safety Management
System Plan Occupational Health Management Measures and Procedures
for Reporting Investigating and Handling of Work Safety Accidents and
Emergency Rescue Plans for Safety Accidents and Sudden Incidents to
strengthen work safety and occupational health management prevent and
reduce production safety accidents and prevent occupational disease
hazards promoting its own sustainable and healthy development. As of the
end of the reporting period the Company's subsidiary Sanjing Mingshui
obtained certification under the ISO 45001 Occupational Health and Safety
Management System.Sanjing Mingshui Occupational Health and Safety
Management System Certification Certificate
9596
Shenglin Asset
Art and Culture
GNC China
Health Technology
Renmintongtai
Marketing Company
Sanjing Daqing
Sanjing Children
Sanjing Qianhe
Sanjing Mingshui
Hapharm Group
Shiyitang Chinese
Medicine
Hayao No.6 Factory
Hayao Sanjing
Hayao General Factory
Technology Center
Beijing Biotechnology
Co. Ltd.Company
headquartersAbout This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
The Company adheres to the safety management principle of "prevention first continuous improvement zero accidents and zero
pollution" promoting a comprehensive safety awareness enhancement plan and building a safety management system with
"leadership taking the lead and all employees participating." This plan integrates case analysis experience sharing ideological
education and humanistic care supported by a reward and penalty mechanism for violations. In 2025 the Company and its
subsidiaries implemented incentive measures such as the Ankang Cup competition striving to create a safety culture of "full
participation and shared responsibility" through diverse cultural promotion.Medal for Advanced Organiza- Medal for Winning Team in the
tion Unit in the Harbin Ankang Harbin Ankang Cup Competition
Cup Competition awarded to awarded to Workshop 301 of
Hayao General Factory Hayao General Factory Case Multi-pronged measures to enhance "dual-hazard" management and empower intrinsic safety
In July 2025 to thoroughly implement the Three-Year Action Plan for Tackling the Root Causes of Production
Safety (2024–2026) issued by the Office of the State Council Work Safety Committee Hayao General Factory
focused on the management of highly toxic easily explosive and other hazardous chemicals and dangerous
operations comprehensively upgrading its "dual-hazard" control level. The Company organized over 40 safety
management personnel and key operational staff to conduct a special seminar on the new version of the Safety
Specifications of Special Work in Hazardous Chemicals Enterprises (GB 30871–2022). Through analysis of typical
cases and benchmarking against regulations the seminar refined approval processes strengthened on-site
supervision and held in-depth discussions on issues such as "insufficient supervisor capabilities" and "difficulties
in dynamic risk control" further solidifying the foundation for on-site management of special operations.This special enhancement initiative aims to shift safety
management towards root cause governance and empower
Medal for Winning Unit in the Medal for Winning Unit in the enterprise intrinsic safety. By transforming institutional
Heilongjiang Province Ankang Harbin Ankang Cup Competi- norms into rigid constraints it has effectively improved the
Cup Competition awarded to tion awarded to Sanchine scientific level of "dual-hazard" control. This is not only an
Sanchine active response to the national campaign for tackling root
causes of work safety but also lays a solid safety foundation
for the Company's high-quality development.Safety-related laws and regulations training
Medal for First Prize in the Team Competi- Certificate for 2024 Suihua City
tion of Mini Fire Stations for Key Fire Occupational Health Enterprise
Safety Units in Daoli District awarded to awarded to Sanjing Mingshui
Shiyitang Chinese Medicine
97 98About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability
Management
Stable Growth Creation Management Shared Future
Case Sanchine building an intelligent safety management system The Company has established a regular risk identification mechanism conducting annual risk identification work and promptly
initiating identification and response actions in the event of significant changes. For identified risks the Company implements
As a comprehensive pharmaceutical enterprise Sanchine is characterized by numerous buildings on its premises classified control: safety risks are categorized into four levels—major significant general and low; environmental and occupation-
and a wide scope of control. Addressing pain points such as cumbersome approvals and blind spots in patrols al health risks are divided into two levels—major and general. Since 2022 the Company and its subsidiaries have progressively
under traditional safety management Sanchine has innovatively built an intelligent safety management system. It carried out risk classification and control work completing the comprehensive identification and assessment of major risks. Corre-
developed a big data approval platform that integrates multiple databases to achieve automatic qualification sponding control measures and emergency plans have been formulated for different risk levels building a dynamically managed
verification and generate assessment reports within three minutes. It upgraded video surveillance to provide risk prevention and control network.
24-hour uninterrupted monitoring and introduced drones for high-altitude inspections. Through the synergy of
"platform + hardware" the efficiency of safety operation approvals increased by 60% hazard investigation cover- In terms of occupational health and safety management the Company set a management goal of zero work safety accidents and
age reached 100% and emergency response speed improved by 50%. This drove the transformation of safety a continuous decline in safety incidents. During the reporting period the Company strictly adhered to the red lines of safety and
management from "passive response" to "proactive prevention" solidifying the safety line of defense in the occupational health management achieving zero work safety accidents fire incidents and traffic accidents. All required occupa-
pharmaceutical sector. tional health examinations were conducted all workplaces with occupational hazard factors were fully monitored and no new
cases of occupational disease were reported providing a solid safety guarantee for production and operations.Key performance
Indicator name Unit 2025 data
Total duration of occupational health and work
safety training for employees Hours 85336
Total duration of occupational health and work
safety training for suppliers and contractors Hours 8226.5
Number of safety drills (fire toxic gas leaks etc.) Number of times 121
Investment in occupational health and safety
management Ten thousand yuan 753.10
Real-time monitoring by the digital system
Investment amount for employee work-related
injury insurance Ten thousand yuan 573.46
Investment amount for employee work safety
liability insurance Ten thousand yuan 6.28
Number of work-related injuries (minor and above) Persons 0
Number of work-related fatalities Persons 0
Number of workdays lost due to work-related
injuries Days 0
Occupational disease incidence rate % 0
Aerial patrols by drones
99 100About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
Rural Revitalization Practices Case Party building as a pen to paint a new picture of revitalization for Sixin Village
To consolidate and expand the achievements of poverty alleviation and effectively link them with rural revitalization the Company The Company has provided targeted assistance to Sixin Village in Yilan County Harbin Heilongjiang Province for
actively responded to the national rural revitalization strategy deployment. Relying on the Heilongjiang Hapharm Foundation it nine consecutive years. Through multi-dimensional initiatives including industrial support infrastructure improve-
planned and implemented the "Hapharm Foundation Rural Guardian Program." Adhering to the principle of "adapting measures to ment healthcare assistance and Party-building collaboration the Company has helped the village achieve poverty
local conditions and implementing targeted policies" the project focuses on health security and implements targeted assistance alleviation and advance rural revitalization. Adopting a systematic and sustainable assistance model the Company
measures empowering rural revitalization through corporate responsibility and effectively safeguarding the health and well-being has aligned its efforts with local needs with Party-building as the guiding approach and industrial empowerment
of the people. During the reporting period the Company donated a total of RMB 60000 worth of common medicines to rural areas. as the key driver leading targeted support initiatives to ensure effective implementation.These supplies were distributed through grassroots health institutions to precisely meet the daily medication and common disease
treatment needs of the residents. The project benefited 16 villages and 4755 people in Heilongjiang Province. The Company’s Party committee has
strengthened rural assistance through
Party-building practices establishing
Case Party-building walls and corridors to Warming old revolutionary base areas and safeguarding health promote Party theories and historical
stories and supporting Sixin Village in being
From October to November 2025 the Company through the Hapharm Foundation visited seven old revolutionary recognized as a provincial-level demonstra-
base area villages in Hulan District Bin County and other areas to conduct health outreach activities contributing tion village for Party-building. The Company
to rural revitalization with practical actions. Based on preliminary surveys of villagers' health needs a total of 9000 has also supported the development of the
boxes of common medicines for colds gastrointestinal issues and inflammation were donated on-site. In collabo- village’s fungus greenhouse industry to
ration with township health centers and village doctors one-on-one medication guidance was provided to villagers increase income for the collective economy
and knowledge on safe medication use was disseminated. Through a model of "material donation + professional and households lifted out of poverty Party building corrido
services" the activity effectively addressed the issues of "difficulty in purchasing and using medicine" for villagers organized a resident work team to assist in
in old revolutionary base areas. It received widespread recognition from local village committees and villagers improving the village environment and used
injecting solid health momentum into rural revitalization. special funds to procure rice flour and
cooking oil to provide support to households
lifted out of poverty during the Spring
Festival. During the reporting period the
Company invested a total of RMB 310200 in
assistance benefiting 630 people. Prior to
the Spring Festival supplies such as rice
flour and cooking oil were distributed to
households lifted out of poverty while
resident team members also assisted in Village-based work team assisting with
cleaning the village environment contribut- sanitation cleanup
ing to rural development and improved
livelihoods.During the reporting period the Company
Invested RMB?????? in rural revitalization,Benefitted????peopleOn-site distribution of medicines
101 102About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Product System for Value
Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation Management Shared Future
The Company has always practiced the volunteer service spirit of "dedication friendship mutual assistance and progress." It
Public Welfare and Social Action innovatively created the distinctive volunteer service project "Hapharm Foundation Rescue is at Hand" focusing on two core areas:
voluntary blood donation and cardiopulmonary resuscitation (CPR) first aid skills training regularly carrying out volunteer service
activities. In 2025 the Company actively organized voluntary blood donation events with 723 volunteers from 15 units participating
to spread warmth. Concurrently two sessions of cardiopulmonary resuscitation (CPR+AED) first aid skills training were conducted
For many years the Company has adhered to the charitable purpose of "developing public welfare and charity and promoting with 90 volunteers enhancing their emergency response capabilities.social civilization and progress." It has deeply integrated public welfare into its corporate sustainability strategy and continuously
invested resources to support the high-quality development of public welfare undertakings. The Company established a profes-
sional and standardized public welfare management system and a full-process support mechanism to ensure that public welfare Case Volunteering to warm the Asian Winter Games: Hapharm Group in action
resources are precisely matched efficiently implemented and practically effective. Over the past three years the Company has
donated a total of RMB 39.394 million in funds and materials benefiting 460400 people. Focusing on key social needs the
Company concentrated on critical areas such as caring for the elderly and children disaster emergency relief and volunteer
During the preparation and hosting of the 9th Asian Winter Games in Harbin the Company actively responded to
services. It meticulously created branded public welfare projects including "Hope Guards Health Hapharm Group Grows with Me"
the call and organized 41 employee volunteers to participate in city-side volunteer services. Volunteers stationed
"Happiness 1+1" "Warming the Sunset Years Guarding the Silver-Haired" and "Hapharm Foundation Rescue is at Hand." This
at Harbin Railway Station provided considerate services such as civility guidance and inquiry resolution for passing
formed a regular diversified and sustainable pattern of public welfare practice demonstrating corporate social responsibility
tourists. Along the torch relay route they cheered for torchbearers with full enthusiasm conveying the warmth of
through practical actions.the Asian Winter Games and demonstrating the demeanor of the host through practical actions. This volunteer
service received wide social recognition. The Social Work Department of the Harbin Municipal Committee of the
During the reporting period the Company Communist Party of China awarded certificates for
city-side volunteer service at the Asian Winter
Games to the 41 volunteers fully affirming their
Invested RMB?????? in the social welfare Helped????people through the public welfare hard work and sense of responsibility. At the
initiatives summary and commendation conference of the
Organized ???employees in volunteer activities Had an average service duration of ?.??hours per Asian Winter Games the Harbin Pharmaceutical
person for employee volunteer activities Group Holding Co. Ltd. Asian Winter Games Volun-
teer Service Team was awarded the provincial-level
"Advanced Collective" title and Hapharm Group.was awarded the municipal-level "Advanced Collec-
Public welfare donations and volunteer services tive" title. These dual honors highlighted the enter-
prises' social responsibility and public welfare
The Company carries out external donations and assistance actions mainly through its Hapharm Foundation and continuously spirit and represented the enterprises' contribu- 9th Harbin Asian Winter Games Torchbearer – Employee
expands forms of public welfare participation by organizing employee volunteer services. tion to the successful hosting of the Asian Winter of Sanjing Pharmaceutical
Games.Case Hapharm Foundation creating a journey of growth through ice and snow study tours
In January 2025 the Company planned and imple-
mented the "Budding Growth: Winter Journey with Ice
and Snow" public welfare study tour activity inviting
children from Daliangshan in Sichuan Yinchuan in
Ningxia and Putian in Fujian to visit Harbin. The
children were immersed in the charm of ice and snow
experienced curling visited art exhibitions and more.This heartwarming initiative lit up childhood dreams
and safeguarded the healthy growth of children. In
May Company volunteers made a special return visit to
Ningxia to fulfill the promises of the "Dream Archives."
Through measures such as holding popular science
lectures on children's bone nutrition and donating
sports equipment they continued to convey care. This
activity not only broadened the horizons of rural Volunteer service activity at Harbin Railway Station
children but also vividly interpreted the public welfare
concept of "Health Guards Dreams" demonstrating
the Company's sense of social responsibility."Budding Growth: Winter Journey with Ice and Snow"
public welfare study tour activity site
103 104About This Message About 2025 Key Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Sustainability
Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth Creation
to Build a Safe and
Management Shared Future
Social services and public value
Case Professional nutrition empowering event services and promoting a healthy lifestyle for all persons
Leveraging its advantages in the pharmaceutical and healthcare industry the Company carried out diverse social service practic-
es focusing on public health services care for special groups and urban public services. While improving service accessibility In May 2025 GNC China participated in the 2025 West Lake Half Marathon in Hangzhou providing participants with
it actively created social value. systematic nutritional support before during and after the race. The Company provided joint health supplements
prior to the race organized support teams to offer on-site supplies during the race and worked with medical and
rehabilitation teams after the race to provide stretching and recovery guidance. An on-site experience area wasCase Travel dialysis: Worry-free love also set up to offer nutrition consultation and health services. Through an integrated “product + service + experi-ence” support model the Company promoted scientific sports nutrition concepts enhanced public awareness of
In December 2025 Heilongjiang Sanjing Nephropathy Hospital healthy dietary supplementation and strengthened its professional image in the sports health sector contributing
centered on patients’ needs integrated health science popular- to a culture of scientific fitness and active lifestyles.ization and service innovation and held a themed event at
Parkson Shopping Center on Central Street. The hospital
assembled a professional medical team of 10 members with
on-site consultation desks case exhibition areas and interac-
tive experience zones. It systematically explained the proce-
dures precautions of travel dialysis as well as off-site dialysis
appointment support services addressing questions and
concerns of kidney disease patients.The event innovatively
integrated traditional Chinese medicine expertise with the
distinctive travel dialysis service enabling patients to under-
stand the feasibility of receiving dialysis while traveling. It
effectively removed travel restrictions for kidney patients and GNC China event support
delivered the humanistic medical philosophy of people-oriented
care. Meanwhile leveraging the large foot traffic of the venue Event Site
the event further raised public awareness of specialized medical
The Company actively fulfilled its social responsibilities focusing on the integrated development of cultural communication and
services bringing high-quality.public welfare. It leveraged its professional resources to carry out diverse cultural activities promoting the combination of cultural
heritage protection and public education and continuously enhancing social and cultural awareness and participation.Case Special exhibition of repatriated cultural relics to promote cultural communication
Case Caring for new employment groups with heartwarming services to safeguard health
In 2025 the Company in collaboration with relevant units held the "National Treasures Gather in the Ice City—
To genuinely care for the physical and mental health of new employment groups such as couriers and ride-hailing Special Exhibition of Repatriated Cultural Relics" in Harbin with multi-party teams participating in the curation and
drivers Renmintongtai actively engaged in safeguarding their health. It took the initiative to integrate its pharma- implementation. Centered on repatriated cultural relics the exhibition presented their historical value and dissemi-
ceutical resources to build a comprehensive health protection system covering medication discounts health nated knowledge on their protection through systematic displays and on-site explanations. The event effectively
services and green medical channels putting into practice the concept of government-enterprise collaboration for enhanced public awareness and consciousness of cultural heritage protection increased social participation and
promoted the integration of cultural resources with the city's cultural tourism. This reflected the enterprise's
public benefit. Renmintongtai selected 500 common responsible values in cultural communication and public welfare practices.drugs to offer exclusive discounts effectively reduc-
ing the medication burden on new employment
groups. It established "Heartwarming Stations" in its
pharmacies providing a range of services including
emergency medicine health monitoring and conve-
nient rest areas. From a cup of plum juice in the
summer to a cup of warming tea in the winter these
small acts of kindness warmed every frontline worker
in new employment groups. Through practical
actions it conveyed warmth helping to enhance the
sense of gain and happiness for these groups and
making the city warmer. Launch ceremony of the event Special Exhibition site Bronze Animal Heads Exhibition
1Heilongjiang Sanjing Nephropathy Hospital is a non-profit Grade III specialized hospital affiliated with Rentongtai (600829.SH)
a subsidiary under the Company’s holding.
105 106About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in
Product System for Value
Group Sustainability Management
Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Stable Growth Creation Management Shared Future
Honors and awards of Hapharm Group
2022.92023.2
HARBIN PHARMACEUTICAL GROUP CO. LTD. was awarded HARBIN PHARMACEUTICAL GROUP CO. LTD.the First "Longjiang was awarded the
Charity Award—Donor Enterprise Award" of the Province "2020–2021 National Blood Donation Promotion Award"
Heilongjiang Civil Administration National Health Commission of the People's Republic of China Red
Information Net Cross Society of China Health Bureau of the Logistic Support
Department of China's Central Military Commission (CMC)
2024.82026.2
HARBIN PHARMACEUTICAL GROUP CO. LTD. Harbin Pharmaceutical Group Co. Ltd. was
was awarded the "Heilongjiang Province Youth Public awarded the Municipal Advanced Collective for
Welfare Activity Research and Practice Base" the 9th Harbin Asian Winter Games
Heilongjiang Provincial Committee of the by the CPC Harbin Municipal Committee and
Communist Youth League Heilongjiang Harbin Municipal People’s Government
Youth Development Foundation
107 108About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth
Creation Management Shared Future
Key Performance Table
Indicator Unit 2025 data Indicator Unit 2025 data
Governance Performance Environmental Performance
Operating revenue RMB 100 million ???.?? Number of participants in environmental management training Person-times ????
Total assets RMB 100 million ???.?? Number of participants in environmental protection publicity and public welfare activities Persons ???
Net assets attributable to shareholders of the listed company RMB 100 million ??.?? Number of emergency drills for environmental risk incidents conducted Sessions ??
Net profit attributable to shareholders of the listed company RMB 100 million ?.?? Number of participants in environmental emergency risk drills Persons ????
Basic earnings per share Yuan per share ?.?? Total greenhouse gas emissions Tonnes of CO2 equivalent ??????.??
Total tax payments RMB 100 million ?.?? Photovoltaic power generation MWh ????.??
Percentage of independent directors % ??.? Proportion of clean energy consumption to total energy consumption % ??.??
Percentage of female directors % ??.? Total clean energy consumption (photovoltaic natural gas) Tonnes of standard coal ?????.??
Number of Board of Directors meetings held Times ?? Direct energy consumption Tonnes of standard coal ?????.??
Director attendance rate % ??? Indirect energy consumption Tonnes of standard coal ?????.??
Number of director training sessions conducted Times ? Total energy consumption Tonnes of standard coal ?????.??
Number of Shareholders' Meetings held Times ? Energy consumption intensity per million RMB in revenue Tonnes/RMB 1 million in revenue ?.??
Total number of investor questions received Times ??? Total water consumption Tonnes ???????
Investor question response rate % ??.?? Freshwater consumption Tonnes ???????
Integrity risk assessment coverage for the Company and its subsidiaries % ??? Recycled water consumption Tonnes ????????
Employee signing rate for compliance commitment letters % ??? Water consumption intensity per million RMB in revenue Tonnes/RMB 1 million in revenue ???.??
Signing rate of Integrity Commitment Letters for bidding suppliers % ??? Total amount of wastewater discharge Tonnes ???????
Number of compliance risks identified during the year Items ??? Chemical oxygen demand (COD) Tonnes ??.??
Number of compliance risk response plans formulated Copies ?? Ammonia nitrogen (NH3-N) Tonnes ?.??
Number of compliance training sessions conducted Times ? Total waste gas Cubic meter ?????.??
Number of employees covered by compliance training Person-times ???? Particulate matter (PM) Tonnes ?.??
Number of anti-commercial bribery and anti-corruption training sessions conducted Times ?? Sulfur oxides (SOx) Tonnes ?.??
Employees participating in anti-commercial bribery and anti-corruption training Person-times ???? Nitrogen oxides (NOx) Tonnes ??.??
Number of internal audit projects conducted Items ?? Volatile organic compounds (VOCs) Tonnes ???.??
Total hazardous waste Tonnes ???.??
Total non-hazardous waste Tonnes ????.??
Indicator Unit 2025 data
Waste recycling rate % ??.??
Environmental Performance
Hazardous waste intensity Tonnes/RMB 1 million in revenue ?.??
Total investment in environmental protection Ten thousand yuan ????.??
Non-hazardous waste intensity Tonnes/RMB 1 million in revenue ?.??
Total duration of environmental management training Hours ????
109 110About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Product System for Value Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth
Creation Management Shared Future
Indicator Unit 2025 data Indicator Unit 2025 data
Social Performance Social Performance
Total R&D investment Ten thousand yuan ?????.?? Number of newly hired employees Persons ???
Proportion of R&D investment to operating revenue % ?.?? Number of ethnic minority employees Persons ???
Proportion of R&D investment to operating revenue of the industrial segment % ?.?? Number of employees with disabilities Persons ???
Proportion of R&D personnel % ??.?? Employee turnover rate % ?
Total number of valid patents held Items ??? Employee benefit coverage rate % ???
Total number of invention patents applied for in the year Items ?? Employee trade union coverage rate % ???
Total number of invention patents granted in the year Items ? Total duration of employee training Hours ??????.?
Total number of valid trademarks held Items ??? Average training hours per person Hours ??.??
Total amount of scientific and technological innovation awards granted Ten thousand yuan ???.?? Total number of training sessions for the year Times ????
Number of projects recognized with scientific and technological innovation awards Pcs ??? Total investment in employee training Ten thousand yuan ???.??
Number of major quality accidents throughout the year Times ? Number of employee training participants Persons ????
Number of non-conformance announcements Items ? Employee training coverage rate % ??
Product qualification rate % ??? Percentage of employees receiving regular performance and career development reviews % ???
Customer feedback handling rate % ??? Total duration of occupational health and work safety training for employees Hours ?????
Number of responsible marketing audits conducted Times ?? Total duration of occupational health and work safety training for suppliers and contractors Hours ????.?
Total number of suppliers Counts ???? Number of safety drills Number of times ???
Proportion of local suppliers % ??.?? Investment in occupational health and safety management Ten thousand yuan ???.??
Local procurement ratio % ??.?? Investment amount for employee work-related injury insurance Ten thousand yuan ???.??
Number of suppliers with document audits conducted Counts ???? Investment amount for employee work safety liability insurance Ten thousand yuan ?.??
Number of suppliers with on-site audits conducted Counts ??? Number of work-related injuries (minor and above) Persons ?
Suppliers that underwent and passed environmental and social audits Counts ?? Number of work-related fatalities Persons ?
Number of supplier training sessions conducted Times ??? Occupational disease incidence rate % ?
Total number of employees Persons ????? Total investment in rural revitalization Ten thousand yuan ??.??
Number of female employees Persons ???? Number of people benefiting from rural revitalization Persons ????
Number of male employees Persons ???? Investment in social welfare Ten thousand yuan ??
Number of employees aged 30 and below Persons ??? Number of people assisted through public welfare initiatives Persons ????
Number of employees aged 31 to 40 Persons ???? Number of employee participations in volunteer activities Person-times ???
Number of employees aged 41 to 50 Persons ???? Average service hours per person for employee volunteer activities Hours ?.??
Number of employees aged 51 and above Persons ???? Number of participants covered by sustainable development training Person-times ??
111 112About This Message About 2025 Key Hapharm Performance in Sustainability
Governance and Compliance Green Operations to Employee Development
Report from the Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon to Build a Safe and Appendix 2025 Sustainability Report
Chairman Group Sustainability Stable Growth
Creation Management Shared Future
Index of Indicators Reader Feedback Form
Guidelines No. 14 of Shanghai Stock Dear ReadersGuidelines for Sustain-
Contents Exchange for Self-Regulation of ability Reports of Listed Companies – Sustainability Chinese Enterprises
Report (Trial) Thank you for reading the 2025 Sustainability Report of HARBIN PHARMACEUTICAL GROUP CO. LTD. We highly value your (CASS-ESG 6.0)
valuable opinions and suggestions and sincerely invite you to evaluate this report. Every piece of your feedback will help us contin-
About This Report - P? uously optimize the quality of our ESG information disclosure improve our sustainability management system better fulfill our
social responsibilities as a pharmaceutical enterprise and safeguard public health and well-being.Message from the Chairman - P?
About Hapharm Group - P?
Sustainability management Communications with stakeholders and due diligence G?.?,G?.?,G?.?I. What is your identity:
Special topic: Empowering with a digital and intelligent
engine building a foundation for high-quality Innovation-driven S?.?
development through technological innovation
Party building leadership and □ Shareholder and investor □ Creditor □ Company employee □ Partner and industry association
responsibility implementation
Governance and
compliance to Construction of corporate
□ Customer □ Supplier and contractor □ Media and public □ Government and regulatory agency
solidify a governance system
G?.?
□ Other __________
foundation for Internal control and compliance
stable growth management
G?.?
Culture of integrity and ethical Anti-commercial bribery anti-corruption and
business practices anti-unfair competition G?.?,G?.? II. Your overall evaluation of this report:Technological r&d and
innovation-driven development Innovation-driven development S?.?
Ethics of science and technolo- Ethics of science and technology What is your evaluation of the overall content of this report:gy
Industrial cooperation and □ Excellent □ Good □ Average □ Poor □ Very poor
Product system exchange
for value creation Product quality and safety Equal treatment to small and medium-sized enterpris-
management es; Safety and quality of products and services S?.?,S?.?Do you think this report comprehensively and objectively reflects the company's actual performance in the environmental social
Customer rights and interests Data security and customer privacy protection
and service assurance S?.? and governance (ESG) fields in 2025
Supply chain management and
security Supply chain security S?.? □ Very adequate □ Relatively adequate □ Average □ Relatively inadequate □ Inadequate
Information security and privacy
protection Data security and customer privacy protection S?.?
Medicine accessibility enhance- Do you think the information disclosed in this report is clear accurate and complete:
ment
Environmental compliance Environmental compliance management E?.? □ Very high □ High □ Average □ Low □ Very lowmanagement
Climate change tackling Climate change tackling E?.?
Green operations What do you think of the structural design and readability of this report:Energy and resource manage-
to advance ment Energy utilization water resource utilization E?.?,E?.?low-carbon □ Excellent □ Good □ Average □ Poor □ Very poor
management Pollutant emissions Pollutant discharge E?.?
Waste management Waste management circular economy E?.?,E?.? III. Your opinions and suggestions:Ecosystem and biodiversity Ecosystem and biodiversity protection E?.?
conservation
Employee rights and well-being S?.?
Talent cultivation and develop- Employees
Employee S?.?ment
development to
build a safe and Occupational health and safety S?.?
shared future
Rural revitalization practices Rural revitalization S?.?
Public Welfare and Social Action Contributions to the society S?.?
113 114About This Message About 2025 Key Sustainability Governance and Compliance Green Operations to Employee Development
Report from the Hapharm Performance in Management Special Topic to Solidify a Foundation for
Product System for Value Advance Low-Carbon Appendix 2025 Sustainability Report
Group Sustainability Stable Growth Creation
to Build a Safe and
Chairman Management Shared Future
Independent Assurance and Statement of Opinion 4. Selected the relevant part of environmental social and governance information in this Report performed analytical assurance procedures for such information confirmed the overall performance situation and evaluated and verified whether the contents
of the Report are complied with.
5. Verified the process management of the principles of inclusiveness substantiality responsiveness and impact in the Report
InFaith Consulting (hereinafter referred to as "InFaith") as entrusted by Harbin Pharmaceutical Group Co. Ltd. (hereinafter and its related AA1000 to confirm the appropriateness of this Statement.referred to as "Hapharm Group") has conducted independent limited assurance of the environmental social and governance 6. Sampled inspection on supporting evidence of data reliability and quality of selected specific performance information.information and data disclosed in 2025 Sustainability Report of Hapharm Group (hereinafter referred to as "the Report") and 7. Recalculated and verified the selected specific performance information.disclosed the assurance result and conclusion to readers of the Report in the form of independent assurance and statement of 8. Performed other procedures that we deemed necessary.opinion.Independence and Capability Assurance Conclusions
1. All the assurance team members of InFaith have no business relationship with Hapharm Group that leads to conflicts of interest. 1. According to the information provided by Hapharm Group and the sample test there is no material misrepresentation in the
The team of InFaith has performed this assurance work independently. Report.
2. The assurance team of InFaith consists of experienced professionals in the industry. Relevant persons have received profes- 2. For the principles of inclusiveness substantiality responsiveness and impact included in AA1000AS v3 the detailed assurance
sional training on the GRI Standards issued by the Global Reporting Initiative AA1000AS v3 ISO 14001 ISO 26000 SA 8000 conclusions are as follows
EcoVadis global supply chain rating and other sustainability-related standards and have adequate understanding and practical
experience in relevant international principles evaluation systems and assurance standards. Hapharm Group has announced major topics that will have substantial influence and impact on the evalua-
Inclusivity tion decision-making actions and performance of the organization and its stakeholders and judged and
Assurance Statement improved the management and performance of the issues.
1. Hapharm Group management shall be fully held accountable for the preparation and content of the Report. It is the responsibili- Hapharm Group has announced significant topics that have a substantial impact and influence on the
ty of InFaith to conduct an assurance based on the scope as described herein and to provide professional assurance opinions to Materiality organization and its stakeholders' assessments decision-making actions and performance. The company
the Report readers and stakeholders. has also conducted judgments and improvements on the management and performance of these topics.
2. Based on the assurance scope limits InFaith shall in accordance with AA1000AS v3 conduct independent limited assurance
and guarantee conclusion for the matters within the scope defined in the Report. In addition to providing independent assurance Hapharm Group has implemented relevant policies and is able to respond to issues that concern the stake-Responsiveness
and making other statements of opinion on the facts of assurance under the conclusion InFaith will not assume any legal or other holders in a timely manner.liabilities for any inquiries for any other purposes or for any other persons who read this Independent Assurance and Statement Hapharm Group has the adequate ability to identify substantive issues. It has implemented balanced and
of Opinion. effective evaluation and disclosure methods and has established a systematic process for monitoring
3. If you have any questions about the contents contained herein or related matters Hapharm Group will reply in a lump sum. Impact measurement evaluation and management impact so as to achieve more efficient management of
4.In the event of any conflict or inconsistency between the Chinese version and the English version of this Independent Assur- decision-making and results within the organization and demonstrate the impact of the substantive issues
ance and Statement of Opinion the Chinese version shall prevail. in an impartial and objective manner.Assurance Standard 3. Based on the procedures implemented and the evidence obtained by InFaith we have found nothing that causes us to doubt
InFaith adopts AA1000AS v3 type-1 Moderate assurance level which includes Hapharm Group's evaluation of the compliance situation the reliability and quality of the specific performance indicators selected in the Report.and degree of the four AA1000AS v3 assurance principles including inclusiveness substantiality responsiveness and impact.Limitation
Assurance Scope The assurance process was carried out at the location within the scope of the Report.Because there is no internationally recognized and commonly used standard for the evaluation and measurement of non-financial
1. The assurance scope is limited to the information and data of Hapharm Group and its affiliated companies covered by the
information the application of different but acceptable information and measurement technologies may affect the comparability
Report excluding the data and information of Hapharm Group suppliers contractors and other third parties.with other institutions.
2. InFaith adopts AA1000AS v3 type-1 Moderate assurance level to evaluate Hapharm Group's compliance nature and degree of
the four assurance principles in AA1000AS v3.If you have any advice for this Independent Assurance and Statement of Opinion you
Assurance Process and Work may contact:
Tel: 021-20740303
In order to gather evidence relevant to the formation of the conclusion InFaith has done the following work: Email: esg@infaith.com.cn
1. Had interviews with the management team and employees of Hapharm Group to understand the overall situation and relevant Address: 17F Tower 1 Lujiazui Riverside Center 257 Lane Binjiang Avenue Pudong
processes of the fulfillment of its environmental social and governance responsibilities. New District Shanghai
2. Verified key developments and policy implementation of the relevant Hapharm Group's organizations and reviewed supporting
evidence for the claims made in the Report. For and on behalf of InFaith Consulting
3. Through the interviews and the assurance of relevant documents learned about Hapharm Group's main stakeholders and their Signed:
expectations and demands the specific communication channels between both parties and how Hapharm Group responds to
their expectations and demands.Shanghai April 2026
115 116HARBIN PHARMACEUTICAL GROUP CO. LTD.
Address: No.7 Qunli Avenue Daoli District Harbin City Heilongjiang Province
Tel:0451-5187 0077
https://www.hayao.com/